Abstract
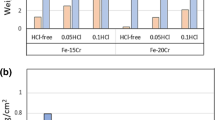
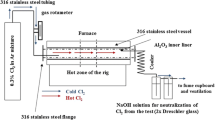
Chloridation-oxidation studies of nine commercial high-temperature alloys were carried out at 800°C in three different H2-HCl-H2O(ν) environments: (A) both low HCl and H2O(ν) partial pressures, where either Cr2O3 or FeCr2O4 is stable, (B) both high HCl and H2O(ν) partial pressures, where FeCrO4 is stable and CrCl2 is metastable, and (C) high HCl partial pressure in the absence of H2O(ν), where either CrCl2 or Cr2O3 is stable. Although alloy 600 has the lowest Cr content of these nine alloys, it showed excellent corrosion resistance in all three environments because of its high Ni content. Alloy 304, with the lowest Ni content of the nine alloys, exhibited poor corrosion resistance in the environment C, fair resistance in the environment B and good resistance in the environment A. Alloy 800 showed very good resistance in Environment A, and fair corrosion resistance in Environment B; however, it suffered linear weight-loss kinetics when exposed to Environment C The alloys 617, 214, HR-160, X, 230 and 86 have good corrosion resistance in Environments A and B because of their relatively high contents of oxide-scale-forming elements Cr and/or Al. In Environment C, chlorine dramatically decreased the adhesion of the scale on the surface, and aluminum in alloys 617 and 214 hardly showed its usual beneficial effect in combating corrosion, nor does silicon in alloy HR-160. Different corrosion mechanisms are proposed for the alloys in the different environments.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Y. Lee and M. J. McNallan,Corrosion,47, 868 (1991).
A. S. Kim and M. J. McNallan,Corroion 46, 746 (1990).
F. H. Stott, R. Prescott, P. Elliott, and M. H. J. H. Al'Atia,High Temp. Technol. 115 (1988).
Y. Y. Lee and M. J. McNallan,Met. Trans. 18A, 1099 (1989).
J. M. Oh, M. J. McNallan, G. Y. Lai, and M. F. Rothman,Met. Trans. 17A, 1087 (1986).
N. S. Jacobson, M. J. McNallan, and Y. Y. Lee,Met. Trans. 17A, 1223 (1986).
M. J. Maloney and M. J. McNallan,Met. Trans. 16B, 751 (1985).
M. J. McNallan and W. W. Liang,Met. Trans. 64, 302 (1981).
N. S. Jacobson,Oxid. Met. 26, 157 (1986).
D. Bramhoff, H. J. Grabke, E. Reese, and H. P. Schmidt,Werkst. Korros. 41, 303 (1990).
K. N. Strafford, P. K. Datta, and G. Forster,Mater. Sci. Engin. A120, 61 (1989).
P. Elliott and G. Marsh.Corros. Sci. 24, 397 (1984).
H. Chu, P. K. Datta, and K. N. Strafford,Oxid. Met. 43, 491 (1995).
K. N. Strafford, P. K. Datta, and G. Forster,Corros. Sci. 29, 703 (1989).
D. Bramhoff, H. J. Grabke, and H. P. Schmidt,Werkst. Korros. 40, 642 (1989).
Y. S. Kim and H. W. Pickering,Metall. Trans. B 13B, 349 (1982).
X.-J. Zheng and R. A. Rapp,Oxid. Met. 48, 527 (1997).
M. W. Chase et al., eds.JANAF Thermodynamic Tables, 3rd Ed. (National Bureau of Standards, 1986).
L. B. Pankratz, J. M. Stuve, and N. A. Gokcen, eds.Thermodynamic Data for Mineral Technology (Bureau of Mines Bulletin 677, 1984).
R. H. Perry and C. H. Chilton, eds.,Chemical Engineers Handbook, 5th Ed. (McGrawHill Book Company, New York, 1973), Chap. 3.
F. N. Mazandarany and R. D. Pehlke,Metall. Trans. 4, 2067 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zheng, X., Rapp, R.A. Chloridation-oxidation of nine commercial high-temperature alloys at 800°C. Oxid Met 48, 553–596 (1997). https://doi.org/10.1007/BF02153465
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02153465