Abstract
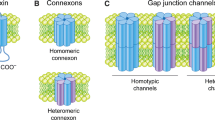
Gap junctions are specialized membrane structures that are involved in the normal functioning of numerous mammalian tissues and implicated in several human disease processes. This mini-review focuses on the regulation of gap junctions through phosphorylation of connexin43 induced by the v-Src or epidermal growth factor receptor tyrosine kinases. These tyrosine kinases markedly disrupt gap junctional communication in mammalian cells. Here, we describe work correlating the alteration of connexin43 function with the ability of the v-Src tyrosine kinase to phosphorylate connexin43 directly on two distinct tyrosine sites in mammalian cells (Y247 and Y265). We also present evidence that proline-rich regions and phosphotyrosine sites of connexin43 may mediate interactions with the SH3 and SH2 domains of v-Src. In contrast to v-Src, the activated epidermal growth factor receptor acts indirectly through activated MAP kinase which may stimulate phosphorylation of connexin43 exclusively on serine. This phosphorylation event is complex because MAP kinase phosphorylates three serine sites in connexin43 (S255, S279, and S282). These findings suggest novel interactions between connexin43, the v-Src tyrosine kinase, and activated MAP kinase that set the stage for future investigations into the regulation of gap junctions by protein phosphorylation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alexandropoulos, K., Cheng, G., and Baltimore, D. (1995).Proc. Natl. Acad. Sci. USA 92, 3110–3114.
Atkinson, M. M., Menko, A. S., Jonson, R. G., Sheppard, J. R., and Sheridan, J. D. (1981).J. Cell Biol. 91, 573–578.
Azarnia, R., and Loewenstein, W. R. (1984).J. Membr. Biol. 82, 191–205.
Azarnia, R., Reddy, S., Kmiecik, T. E., Shalloway, D., and Loewenstein, W. R. (1988).Science 239, 398–401.
Berridge, M. J. (1987).Annu. Rev. Biochem. 56, 156–194.
Berthoud, V. M., Ledbetter, M. L. S., Hertzberg, E. L., and Saez, J. C. (1992).Eur. J. Cell Biol. 57, 40–50.
Beyer, E. C. (1993).In. Rev. Cytol. 137C, 1–37.
Bignami, M., Rosa, S., Falcone, G., Tato, F., Katoh, F., and Yamasaki, H. (1988).Mol. Carcinog. 1, 67–75.
Brisette, J. L., Kumar, N. M., Gilula N. B., and Dotto, G. P. (1991).Mol. Cell Biol. 11, 5364–5371.
Chang, C.-C., Trosko, J. E., Kung, H.-J., Bombick, D., and Matsumura, F. (1985).Proc. Natl. Acad. Sci. USA 82, 5360–5364.
Cobb, B. S., Schaller, M. D., Leu, T.-H., and Parsons, J. T. (1994).Mol. Cell Biol. 14, 147–155.
Crow, D. S., Beyer, E. C., Paul, D. L., Kobe, S. S., Lau, A. F. (1990).Mol. Cell Biol. 10, 1754–1763.
Crow, D. S., Kurata, W. E., and Lau, A. F. (1992).Oncogene 7, 999–1003.
Filson, A. J., Azarnia, R., Beyer, E. C., Loewenstein, W. R., and Brugge, J. S. (1990).Cell Growth Differ. 1, 661–668.
Goldberg, G. S., and Lau, A. F. (1993).Biochem. J. 295, 735–742.
Guan, J.-L. and Shalloway, D. (1992).Nature 358, 690–692.
Gupta, S. K., Gallego, C., Johnson, G. L., and Heasley, L. E. (1992).J. Biol. Chem 267, 7987–7990.
Jou, Y.-S., Layhe, B., Matesic, D. F., Chang, C.-C., de Feijter, A. W., Lockwood, L., Welsch, C. W., Klaunig, J. E., and Trosko, J. E. (1995).Cacinogenesis 16, 311–317.
Kamps, M. P., Buss, J. E., and Sefton, B. M. (1985).Proc. Natl. Acad. Sci. USA 82, 4625–4628.
Kanemitsu, M. Y., and Lau, A. F. (1993).Mol. Biol. Cell 4, 837–848.
Kefalas, P., Brown, T. R. P., and Brickell, P. M. (1995).Int. J. Biochem. Cell Biol. 27, 551–563.
Kurata, W. E., and Lau, A. F. (1994).Oncogene 9, 329–335.
Lau, A. F., Kanemitsu, M. Y., Kurata, W. E., Danesh, S., and Boynton, A. L. (1992).Mol. Biol. Cell 3, 865–874.
Lampe, P. D. (1994).J. Cell Biol. 127, 1895–1905.
Loo, L. W. M., Berestecky, J. M., Kanemitsu, M. Y., and Lau, A. F. (1995).J. Biol. Chem. 270, 12751–12761.
Madhukar, B. V., Oh, S. Y., Chang, C. C., Wade, M., and Trosko, J. E. (1989).Carcinogenesis 10, 13–20.
Malarkey, K., Belham, C. M., Paul, A., Graham, A., McLees, A., Scott, P. H., and Plevin, R. (1995).Biochem. J. 309, 361–375.
Maldonado P. E., Rose, B., and Loewenstein, W. R. (1988).J. Membr. Biol. 106, 203–210.
Marengere, L. E. M., and Pawson, T. (1994).J. Cell Sci. 18, 97–104.
Musil, L. S. (1994). InMolecular Mechanisms of Epithelial Cell Junctions: From Development to Disease: Structure and Assembly of Gap Junctions (Citi, S., ed.), R. G. Landes Co., Austin, pp. 173–194.
Musil, L. S., and Goodenough, D. A. (1991).J. Cell Biol. 115, 1357–1374.
Musil, L. S., Cunningham, B. A., Edelman, G. M., Goodenough, D. A. (1990).J. Cell Biol. 111, 2077–2088.
Nishizuka, Y. (1988).Nature 334, 661–665.
Oh, S. Y., Grupen, C. G., and Murray, A. W. (1991).Biochim. Biophys. Acta 1094, 243–245.
Parsons, J. T., and Weber, M. J. (1989).Curr. Top. Microbiol. Immunol. 147, 79–127.
Rozakis-Adcock, M., McGlade, J., Mbamalu, G., Pelicci, G., Daly, R., Li, W., Batzer, A., Thomas, S., Brugge, J., Pelicci, P.G., Schlessinger, J., and Pawson, T. (1992).Nature 360, 689–692.
Saez, J. C., Berthoud, V. M., Moreno, A. P., and Spray, D. C. (1993). InAdvances in Second Messenger and Phosphoprotein Research: Gap Junctions (Shenolikar, S., and Nairn, A. C., eds.), Raven Press, New York, pp. 163–198.
Seger, R., and Krebs, E. G. (1995).FASEB J. 9, 726–735.
Smith, M. R., DeGudicibus, S. J., and Stacy, D. W. (1986).Nature 320, 540–543.
Spray, D. C. (1994). InMolecular Mechanisms of Epithelial Cell Junctions: From Development to Disease: Structure and Assembly of Gap Junctions (Citi, S., ed.), R. G. Landes Co., Austin, pp. 195–215.
Stagg, R. B., and Fletcher, W. H. (1990).Endocrine Rev. 11, 302–325.
Superti-Furga, G., and Courtneidge, S. A. (1995).BioEssays 17, 321–330.
Swenson, K. I., Piwnica-Worms, H., McNamee, H., and Paul, D. L. (1990).Cell Regul. 1, 989–1002.
Warn-Cramer, B. J., Lampe, P. D., Kurata, W. E., Kanemitsu, M Y., Loo, L. W. M., Eckhart, W., and Lau, A. F. (1996).J. Biol. Chem. 271, 3779–3786.
Willingham, M. C., Jay, G., and Pastan, I. (1979).Cell 18, 125–134.
Wolburg, H., and Rohlmann, A. (1995).Int. Rev. Cytol. 157, 315–373.
Zang, Q., Frankel, P., and Foster, D. A. (1995).Cell Growth Differ. 6, 1367–1373.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lau, A.F., Kurata, W.E., Kanemitsu, M.Y. et al. Regulation of connexin43 function by activated tyrosine protein kinases. J Bioenerg Biomembr 28, 359–368 (1996). https://doi.org/10.1007/BF02110112
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02110112