Abstract
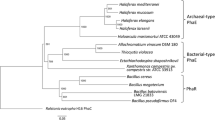
Four polyhydroxyalkanoate (PHA) depolymerases were purified from the culture fluid ofPseudomonas lemoignei: poly(3-hydroxybutyrate) (PHB), depolymerase A (M r , 55,000), and PHB depolymerase B (M r , 67,000) were specific for PHB and copolymers of 3-hydroxybutyrate (3HB) and 3-hydroxyvalerate (3HV) as substrates. The third depolymerase additionally hydrolyzed poly(3-hydroxyvalerate) (PHV) at high rates (PHV depolymerase;M r , 54,000). The N-terminal amino acid sequences of the three purified proteins, of a fourth partially purified depolymerase (PHB depolymerase C), and of the PHB depolymerases ofComamonas sp. were determined. Four PHA depolymerase genes ofP. lemoignei (phaZ1,phaZ2,phaZ3, andphaZ4) have been cloned inEscherichia coli, and the nucleotide sequence ofphaZ1 has been determined recently (D. Jendrossek, B. Müller, and H. G. Schlegel,Eur. J. Biochem. 218, 701–710, 1993). In this study the nucleotide sequences ofphaZ2 andphaZ3 were determined.PhaZ1,phaZ2, andphaZ4 were identified to encode PHB depolymerase C, PHB depolymerase B, and PHV depolymerase, respectively.PhaZ3 coded for a novel PHB depolymerase ofP. lemoignei, named PHB depolymerase D. None of the four genes harbored the PHB depolymerase A gene, which is predicted to be encoded by a fifth depolymerase gene ofP. lemoignei (phaZ5) and which has not been cloned yet. The deduced amino acid sequences ofphaZ1–phaZ3 revealed high homologies to each other (68–72%) and medium homologies to the PHB depolymerase gene ofAlcaligenes faecalis T1 (25–34%). Typical leader peptide amino acid sequences, lipase consensus sequences (Gly-Xaa-Ser-Xaa-Gly), and unusually high proportions of threonine near the C terminus were found in PhaZ1, PhaZ2, and PhaZ3. Considering the biochemical data of the purified proteins and the amino acid sequences, PHA depolymerases ofP. lemoignei are most probably serine hydrolases containing a catalytical triad of Asp, His, and Ser similar to that of lipases. A comparison of biochemical and genetic data of various eubacterial and one eukaryotic PHA depolymerases is provided also.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
A. J. Anderson and E. A. Dawes (1990)Microbiol. Rev. 54 450–472.
H. Blaak, J. Schnellmann, S. Walter, B. Henrissat, and H. Schrempf (1993)Eur. J. Biochem. 214 659–669.
H. Blum, H. Beier, and H. J. Gross (1987)Electrophoresis 8 93–99.
B. H. Briese, D. Jendrossek, and H. G. Schlegel (1994)FEMS Microbiol. Lett. 117 107–112.
C. L. Brucato and S. S. Wong (1991)Arch. Biochem. Biophys. 290 497–502.
K. P. Budwill, M. Fedorak, and W. J. Page (1992)Appl. Environ. Microbiol. 58 1398–1401.
A. A. Chowdhury (1963)Arch. Mikrobiol. 47 167–200.
F. P. Delafield, K. E. Cooksey, and M. Doudoroff (1965)J. Biol. Chem. 240 4023–4028.
J. Devereux, P. Haeberli, and O. Smithies (1984)Nucleic Acids Res. 12 387–395.
Y. Doi (1990)Microbial Polyesters, VHC, New York.
Y. Doi, Y. Kanesawa, M. Kunioka, and T. Saito (1990)Macromolecules 23 26–31.
L. F. Fägerstam, G. Pettersson, and J. Å. Engström (1984)FEBS Lett. 167 309–315.
R. D. Fields, F. Rodriguez, and R. K. Finn (1974)J. Appl. Polym. Sci. 18 3571–3579.
W. G. C. Forsyth, A. C. Hayward, and R. B. Roberts (1958)Nature (London) 182 800–801.
T. Fukui, K. Narikawa, Y. Miwa, T. Shirakura, T. Saito, and K. Tomita (1988)Biochim. Biophys. Acta 952 164–171.
R. C. Fuller, J. P. O'Donnell, J. Saulnier, T. E. Redlinger, J. Forster, and R. W. Lenz (1992)FEMS Microbiol. Rev. 103 279–288.
H. J. Gilbert, J. Hall, G. P. Hazlewood, and L. M. A. Ferreira (1990)Mol. Microbiol. 4 759–767.
N. R. Gilkes, M. L. Langsford, D. G. Kilburn, R. V. Miller, Jr., and R. A. J. Warren (1984)J. Biol. Chem. 259 10455–10459.
N. R. Gilkes, B. Henrissat, D. G. Kilburn, R. C. Miller, Jr., and R. A. J. Warren (1991)Microbiol. Rev. 55 303–315.
I. J. Goldstein and C. E. Hayes (1978)Adv. Carbohydr. Chem. Biochem. 35 127–140.
O. Grépinet, M.-C. Chebrou, and P. Béguin (1988)J. Bacteriol. 170 4582–4588.
E. K. Gum and R. D. Brown (1976)Biochim. Biophys. Acta 446 371–386.
J. Hall, G. P. Hazlewood, N. S. Huskisson, A. J. Durrant, and H. J. Gilbert (1989)Mol. Microbiol. 3 1211–1219.
H. Hippe and H. G. Schlegel (1967)Arch. Mikrobiol. 56 278–299.
G. W. Huisman, E. Wonink, R. Meima, B. Kazemier, P. Terpstra, and B. Witholt (1991)J. Biol. Chem. 266 2191–2198.
K. E. Jäger, S. Ransac, H. B. Koch, F. Ferrato, and B. W. Dijkstra (1993)FEBS Lett. 332 143–149.
P. H. Janssen and C. G. Harfoot (1990)Arch. Microbiol. 154 253–259.
D. Jendrossek, I. Knoke, R. B. Habibian, A. Steinbüchel, and H. G. Schlegel (1993)J. Environ. Polym. Degrad. 1 53–63.
D. Jendrossek, B. Müller, and H. G. Schlegel (1993)Eur. J. Biochem. 218 701–710.
R. Kavelman and B. Kendrick (1978)Mycologia 70 87–103.
W. M. Konyecsni and V. Deretic (1990)J. Bacteriol. 172 2511–2520.
U. K. Laemmli (1970)Nature (London) 227 680–685.
J. Lechner and F. Wieland (1989)Annu. Rev. Biochem. 58 173–194.
H. Lis and N. Sharon (1993)Eur. J. Biochem. 218 1–27.
C. J. Lusty and M. Doudoroff (1966)Proc. Natl. Acad. Sci. USA 56 960–965.
M. Matavulj and H. P. Molitoris (1992)FEMS Microbiol. Rev. 103 323–331.
D. W. McLellan and P. J. Halling (1988)FEMS Microbiol. Lett. 52 215–218.
A. Meinke, C. Braun, N. R. Gilkes, D. G. Kilburn, R. C. Miller, Jr., and R. A. J. Warren (1991)J. Bacteriol. 173 308–314.
J. Mergaert, C. Anderson, A. Wouters, J. Swings, and K. Kersters (1992)FEMS Microbiol. Rev. 103 317–321.
J. M. Merrick (1965)J. Bacteriol. 90 965–969.
J. M. Merrick and M. Doudoroff (1964)J. Bacteriol. 88 60–71.
J. M. Merrick, G. Lundgren, and R. M. Pfister (1965)J. Bacteriol. 89 234–239.
B. Müller and D. Jendrossek (1993)Appl. Microbiol. Biotechnol. 38 487–492.
K. Mukai and Y. Doi (1993) in H. G. Schlegel and A. Steinbüchel (Eds.),Proceedings of the International Symposium on Bacterial Polyhydroxyalkanoates, Goltze Druck, Göttingen, pp. 457–458.
K. Nakayama, T. Saito, T. Fukui, Y. Shirakura, and K. Tomita (1985)Biochim. Biophys. Acta 827 63–72.
C. L. Oakley (1971)Meth. Microbiol. 5A 173–218.
F. W. Paradis, H. Zhu, P. J. Krell, J. P. Phillips, and C. W. Forsberg (1993)J. Bacteriol. 175 7666–7672.
B. Persson, G. Bentsson-Olivecrona, S. Enerback, T. Olivecrona, and H. Jornvall (1989)Eur. J. Biochem. 179 39–45.
T. Platt (1986)Annu. Rev. Biochem. 55 339–372.
A. P. Pugsley and M. Schwartz (1985)FEMS Microbiol. Rev. 32 3–38.
T. Saito, K. Suzuki, J. Yamamoto, T. Fukui, K. Miwa, K. Tomita, S. Nakanishi, S. Odani, J.-I. Suzuki, and K. Ishikawa (1989)J. Bacteriol. 171 184–189.
T. Saito, H. Saegusa, Y. Miyata, and T. Fukui (1992)FEMS Microbiol. Rev. 103 333–338.
T. Saito, A. Iwata, and T. Watanabe (1993)J. Environm. Polym. Degrad. 1 99–105.
J. Sambrook, E. F. Fritsch, and T. Maniatis (1989)Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
F. Sanger, S. Nicklen, and A. R. Coulson (1977)Proc. Natl. Acad. Sci. USA 74 5463–5467.
A. Schirmer, D. Jendrossek, and H. G. Schlegel (1993)Appl. Environ. Microbiol. 59 1220–1227.
H. G. Schlegel, G. Gottschalk, and R. v. Bartha (1961)Nature (London) 191 463–465.
H. G. Schlegel, H. Kaltwasser, and G. Gottschalk (1961)Arch. Mikrobiol. 117 475–481.
H. G. Schlegel and G. Gottschalk (1962)Angewandte Chem. 74 342–347.
J. D. Schrag, L. Yunge, S. Wu, and M. Cygler (1991)Nature 351 761–764.
J. P. Segrest and R. L. Jackson (1972)Methods Enzymol. 28 54–63.
S. Shoemaker, V. Schweickart, M. Ladner, D. Gelfand, S. Kwok, K. Myambo, and M. Innis (1983)Bio/Technology 1 691–696.
H. Stegemann, H. Francksen, and V. Macko (1973)Z. Naturforsch. 28c 722–732.
A. Steinbüchel (1991) in D. Byrom (Ed.),Biomaterials, Macmillan Press, London, pp. 123–213.
A. Steinbüchel, E. Hustede, M. Liebergesell, U. Pieper, A. Timm, and H. Valentin (1992)FEMS Microbiol. Rev. 103 217–230.
A. Steinbüchel, E.-M. Debzi, R. H. Marchessault, and A. Timm (1993)Appl. Microbiol. Biotechnol. 39 443–449.
B. Svensson, K. Larsen, I. Svendsen, and E. Boel (1983)Carsberg Res. Commun. 48 529–544.
T. Tanio, T. Fukui, Y. Shirakura, T. Saito, K. Tomita, T. Kaiho, and S. Masamune (1982)Eur. J. Biochem. 124 71–77.
T. T. Teeri, P. Lehtovaara, S. Kauppinnen, I. Salovuori, and J. Knowles (1987)Gene 51 43–52.
A. Timm and A. Steinbüchel (1992)Eur. J. Biochem. 209 15–30.
H. Towbin, T. Staehelin, and J. Gordon (1979)Proc. Natl. Acad. Sci. USA 76 4350–4354.
H. Van Tilbeurgh, P. Tomme, M. Claeyssens, R. Bhikhabhai, and G. Pettersson (1986)FEBS Lett. 204 223–227.
T. Watanabe, K. Suzuki, W. Oyanagi, K. Ohnishi, and H. Tanaka (1990)J. Biol. Chem. 265 15659–15665.
M. E. E. Watson (1984)Nucl. Acids Res. 12 5145–5164.
F. K. Winkler, A. D'Acry, and W. Hunziker (1990)Nature 343 771–774.
S. Wohlfarth, C. Hoesche, C. Strunk, and U. K. Winkler (1992)J. Gen. Microbiol. 138 1325–1335.
E. Yagüe, P. Béguin, and J.-A. Aubert (1990)Gene 89 61–67.
K. Yamada, K. Mukai, and Y. Doi (1993)Int. J. Biol. Macromol. 15 215–220.
K. Yanai, N. Takaya, N. Kojima, H. Horiuchi, H. Ohta, and M. Takagi (1992)J. Bacteriol. 174 7398–7406.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Briese, B.H., Schmidt, B. & Jendrossek, D. Pseudomonas lemoignei has five poly(hydroxyalkanoic acid) (PHA) depolymerase genes: A comparative study of bacterial and eukaryotic PHA depolymerases. J Environ Polym Degr 2, 75–87 (1994). https://doi.org/10.1007/BF02074776
Issue Date:
DOI: https://doi.org/10.1007/BF02074776