Abstract
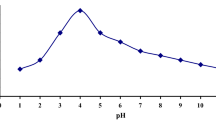
Adsorption behavior of zinc ions on hydrous zirconium oxide (HZO) in aqueous solution has been studied as a function of concentration (10−2–10−8M), temperature (303–333 K) and pH 3–8 of adsorptive solution applying radiotracer technique. The kinetics of adsorption follows first order rate law and agrees well with the classical Freundlich isotherm in the entire range of adsorptive concentration. The removal was found to be increasing with pH of the adsorptive solution while it was suppressed in the presence of acid concentrations. The overall process is found to be endothermic and irreversible in nature.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
B. A. BROWN, A. A. BROWN, G. C. LALOR, Environ. Geochem. Health, 17 (1995) 545.
K. L. TOTAWAT, R. N. UPADHYAY, S. C. CHAUHAN, Ind. J. Environ. Health, 36 (1994) 237.
A. SHARMA, M. SHARMA, J. Environ. Biol., 15 (1994) 221.
V. N. IYER, R. SARIN, Asian Environ., 11 (1989) 39.
S. MUSIC, M. RISTIC, J. Radioanal. Nucl. Chem., 120 (1988) 289.
H. KAWANO, Y. NAKAI, T. MATSUDA, T. NAGAI, Talanta, 33 (1986) 191.
S. MUSTAFA, M. SAFDAR, G. NAWAB, Phys. Chem., 9 (1990) 79.
M. BRANDAO, B. SUELI, F. GALEMBECK, Colloids Surf., 48 (1990) 351.
S. MUSIC, M. RISTIC, J. Radioanal. Nucl. Chem., 162 (1992) 351.
S. M. HASANY, F. A. CHUGHTAI, A. GHAFFAR, Sep. Sci. Technol., 26 (1991) 1131.
G. MICERA, C. GESSA, P. MELIS, A. PREMOLI, R. DALLOCHIO, S. DEIANA, Colloids. Surf., 17 (1986) 386.
S. MUSTAFA, M. SAFDAR, S. Y. HUSSAIN, J. Surf. Sci. Technol., 5 (1989) 267.
H. TAMURA, M. NAGAYAMA, Prog. Batt. Sol. Cells, 5 (1984) 143.
M. RASHID, M. EJAZ, Appl. Radiation Isotopes, 37 (1986) 501.
S. P. MISHRA, V. K. SINGH, D. TIWARI, Appl. Radiation Isotopes, 47 (1995) 15.
H. S. MAHAL, B. VENKATRAMANI, K. S. VENKATESWARLU, J. Inorg. Nucl. Chem., 43 (1981) 3335.
J. A. GLADSDEN, Infrared Spectra of Minerals and Related Compounds, Butterworths, London, 1975.
S. P. MISHRA, N. SRINIVASU, D. TIWARY, Appl. Radiation Istopes, 43 (1992) 1253.
P. BENES, V. MAJER, Trace Chemistry of Aqueous Solution, Elsevier, Amsterdam, 1980.
F. HELFFERICH, Ion Exchange, McGraw Hill, New York, 1962.
M. OKAZAKI, K. TAKAMIDOH, I. YAMANE, Soil Sci. Plant Nutr., 32 (1986) 523.
G. A. PARKS, Chem. Rev., 65 (1965) 177.
B. VOLESKY, FEMS Microbiol. Rev., 14 (1994) 291.
R. FUJIYOSHI, A. S. EUGENE, M. KATAYAMA, Appl. Radiation Isotopes, 43 (1992) 1223.
R. J. ZASOSKI, R. G. BURAU, Soil Sci. Soc. Am. J., 52 (1986) 81.
C. E. COWAN, J. M. ZACHARA, C. T. RESCH, Environ. Sci. Technol., 25 (1991) 437.
A. R. BOWERS, C. P. HUANG, J. Colloids Interface Sci., 105 (1985) 197.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mishra, S.P., Singh, V.K. & Tiwari, D. Inorganic particulates in removal of toxic heavy metal ions. Journal of Radioanalytical and Nuclear Chemistry Articles 210, 207–217 (1996). https://doi.org/10.1007/BF02055419
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02055419