Summary
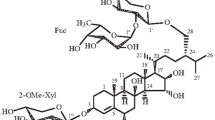
On the basis of comparative chemical and spectral data, the structure of the major saponin, luzonicoside, from the starfishEchinaster luzonicus has been elucidated as2. This is a further example of a novel class of steroidal cyclic glycoside from starfish of the genusEchinaster. Its structure includes a Δ7, 3β,6β-dioxygenated-23-oxosteroidal moiety, already found in the saponins ofEchinaster sepositus, and a trisaccharide moiety, β-D-galactopyranosyl-(1→2) α-L-arabinopyranosyl-(1→2)-β-D-glucuronopyranosyl, bridging C-3 and C-6 of the steroid.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
F. De Simone, A. Dini, E. Finamore, L. Minale, C. Pizza, R. Riccio and F. Zollo, J. chem. Soc. Perkin I,1981, 1855.
L. Minale, R. Riccio, F. De Simone, A. Dini, C. Pizza and E. Ramundo, Tetrahedron Lett.1978, 2609.
R. Riccio, F. De Simone, A. Dini, L. Minale, C. Pizza, F. Senatore and F. Zollo, Tetrahedron Lett.1981, 1557.
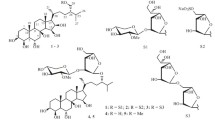
HPLC-chromatograms ofE. sepositus andE. luzonicus saponins are almost indistinguishable. Both chromatograms contained 3 peaks of which the central one corresponded to the major component. In theE. luzonicus it accounts for more than 90% of the total saponin content. The 2 minor saponins were obtained still in admixture with the major one and, in analogy with the minor saponins ofE. sepositus, on acid hydrolysis, they gave 24-nor-22(R)-chloro-5α-cholesta-8,14-diene-3β,23(S)-diol (more polar saponin) and 22(R)-chloro-5α-cholesta-8,14-dien-3β,23(S)-diol along with minor amounts of 27-nor-24-methyl-22(R)-chloro-5α-cholesta-8,14-dien-3β,23(S)-diol (less polar saponin)13. The13C-NMR spectra of both mixtures confirmed the origin of these chlorohydrins from their corresponding epoxides (part-structures5–7).
B. Lindbery, in: Methods in Enzymology, vol. 28, part B, p. 178. Ed. V. Ginsburg. Academic Press, New York and London 1972.
W. Voelter, E. Breitmaier, E.B. Rathbone and A.M. Stephen, Tetrahedron29, 3845 (1973).
E. Breitmaier and W. Voelter, Tetrahedron29, 227 (1973).
L. Minale, R. Riccio, F. De Simone, A. Dini and C. Pizza, Tetrahedron Lett.1979, 645.
Author information
Authors and Affiliations
Additional information
This contribution is part of the Progetto Finalizzato ‘Oceanografia e Fondi Marini’ del C.N.R., Roma.
Acknowledgments. We wish to thank Mrs M. Pusset, Laboratoire des Plantes Médicinales du C.N.R.S., Nouméa, for the extraction of starfish and the divers of the Centre Q.R.S.T.O.M. de Nouméa for collection.
Rights and permissions
About this article
Cite this article
Riccio, R., Dini, A., Minale, L. et al. Starfish saponins VII. Structure of luzonicoside, a further steroidal cyclic glycoside from the pacific starfishEchinaster luzonicus. Experientia 38, 68–70 (1982). https://doi.org/10.1007/BF01944532
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01944532