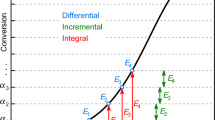
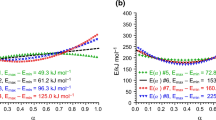
Abstract
It is known that the application of an inappropriate kinetic method to the thermal analysis of complex systems can lead to misleading results. To avoid this problem, the new parameter ‘instantaneous mean activation energy’ is introduced and the Ozawa-Flynn-Wall method is assessed as a means of obtaining it. It is concluded that good results can generally be obtained by this method, provided that the various reactions occurring in the complex system are of the same type. The pyrolysis of coal is considered as a possible application of the method.
Zusammenfassung
Es ist bekannt, da\ die Anwendung einer ungeeigneten kinetischen Methode bei der thermischen Analyse komplexer Systeme zu falschen Ergebnissen führen kann. Um dieses Problem zu vermeiden, wird der neue Parameter „momentane mittlere Aktivierungsenergie“ eingeführt. Die Ozawa-Flynn-Wall-Methode wird als zur Bestimmung dieses Parameters geeignet angesehen. Es wird der Schlu\ gezogen, da\ mit dieser Methode im allgemeinen gute Ergebnisse zu erhalten sein sollten, vorausgesetzt, da\ die in komplexen Systemen verlaufenden verschiedenen Reaktionen vom gleichen Typ sind. Die Pyrolyse von Kohle wird als mögliche Anwendung dieser Methode angesehen.
РЕжУМЕ
пРИМЕНЕНИЕ НЕсООтВЕ тстВУУЩЕгО кИНЕтИЧЕ скОгО МЕтОДА к тЕРМИЧЕскОМ У АНАлИжУ слОжНых сИстЕМ МОжЕт пРИВЕстИ к ОшИБОЧНыМ РЕжУльтАтАМ. Дль РЕшЕ НИь ЁтОИ пРОБлЕМы ВВОДИтсь НОВыИ пАРАМ ЕтР «МгНОВЕННАь сРЕД Ньь ЁНЕРгИь АктИВАцИИ» И МЕтОД ОжАВА-ФлИННА-ВАллА Ис пОльжОВАН Дль пОлУЧЕ НИь ЁтОгО пАРАМЕтРА. сДЕлАНО жА клУЧЕНИЕ, ЧтО В ОБЩЕМ, ЁтИМ МЕтОДОМ МО гУт Быть пОлУЧЕНы хОР ОшИЕ РЕжУльтАты, НО пРИ Усл ОВИИ, ЧтО пРОтЕкАУЩИЕ В слОжНО И сИстЕМЕ РЕАкцИИ ьВл ьУтсь ОДНОгО И тОгО жЕ тИпА. с ЧИтАЕтсь, ЧтО ЁтОт МЕтОД МОжЕт Быть пРИМ ЕНЕН Дль пИРОлИжА Угл ь.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- a :
-
Parameter used in the Doyle approximation (Eq. (3))
- A :
-
Arrhenius pre-exponential factor
- b :
-
Parameter used in the Doyle approximation (Eq. (3))
- c i :
-
Contribution of an individual reaction to the overall reaction
- E inst :
-
Instantaneous mean activation energy (Eq. (13))
- E OFW :
-
Activation energy as calculated by the Ozawa-Flynn-Wall method
- f(α) :
-
Function describing the dependence of the rate constant on the degree of conversion
- F(α) :
-
Integral of 1/ f(α) (Eq. (2))
- i(subscript):
-
Denotes a component reaction
- R :
-
Gas constant
- t :
-
Time
- T :
-
Temperature
- v :
-
Reaction rate
- α :
-
Degree of conversion
- Β :
-
Heating rate
- Μ 2E :
-
Instantaneous second moment of activation energies (Eq. (18))
References
A. W. Czanderna, J. R. Biegen and W. Kollen, J. Colloid. Interface Sci., 34 (1970) 406.
Y. Tokoro, T. Uchijima and Y. Yoneda, J. Catal., 37 (1975) 44.
T. Ozawa, Bull. Chem. Soc. Japan, 38 (1965) 1881.
J. H. Flynn and L. A. Wall, Polym. Lett., 4 (1966) 323.
J. H. Flynn, J. Thermal Anal., 27 (1983) 95.
M. A. Serageldin and Wei-ping Pan, Thermochimica Acta, 71 (1983) 1.
H. Juentgen and K. H. van Hcek, Fortschr. Chem. Forsch, 13 (1970) 601.
J. P. Elder, J. Thermal Anal., 29 (1984) 1327.
D. B. Anthony and J. B. Howard, AlCHE, J., 22 (1976) 625.
J. J. Reuther, R. D. Daley, J. J. Warchol and J. A. Withum, Fuel, 63 (1984) 604.
P. R. Solomon, D. G. Hamblen, R. M. Carangelo, J. R. Markham and M. D. DiTaranto, Prepr. Amer. Chem. Soc. Div. Fuel Chem., 29 (1984) 83.
E. M. Suuberg, W. A. Peters and J. B. Howard, Ind. Eng. Chem. Process Des. Dev.,. 17 (1978) 37.
J. B. Howard, Chemistry of Coal Utilization, Second Supplementary Volume, M. A. Elliot (Ed.), Wiley-Interscience, New York, 1981, p. 727.
D. B. Anthony, J. B. Howard, H. C. Hottel and H. P. Meissner, 15th Int. Symp. Combustion, 1974, p. 1303.
C. D. Doyle, J. Appl. Polymer Sci., 6 (1962) 639.
J. W. Cumming, Fuel, 63 (1984) 1436.
J. H. Flynn, Thermochim. Acta, 37 (1980) 225.
T. Ozawa, J. Thermal Anal., 7 (1975) 601.
K. M. Sprouse and M.D. Schuman, Combust. Flame, 43 (1981) 265.
D. B. Anthony, J. B. Howard, H. C. Hottel and H. P. Meissner, Fuel, 55 (1976) 121.
Page 739 of ref. 13.
V. Koch, H. Juentgen and W. Peters, Brennstoff-chemie, 50 (1969) 369.
Author information
Authors and Affiliations
Additional information
The author wishes to thank the British Gas Corporation for permission to publish this work.
Rights and permissions
About this article
Cite this article
Dowdy, D.R. Meaningful activation energies for complex systems. Journal of Thermal Analysis 32, 137–147 (1987). https://doi.org/10.1007/BF01914556
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01914556