Summary
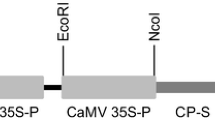
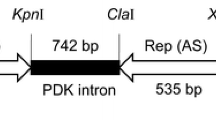
Generation of transgenic papaya (Carica papaya L.) has been hampered by the low rates of transformation achieved by conventionalAgrobacterium infection or microprojectile bombardment. We describe an efficientAgrobacterium-mediated transformation method based on wounding of cultured embryogenic tissues with carborundum in liquid phase. Embryogenic tissues were obtained from cultured immature zygotic embryos collected 75–90 days after pollination. The expressible coat protein (CP) gene of a Taiwan strain of papaya ringspot virus (PRSV) was constructed in a Ti binary vector pBGCP, which contained the NPT-II gene as a selection marker. The embryogenic tissues were vortexed with 600 mesh carborundum in sterile distilled water for 1 min before treating with the disarmedA. tumefaciens containing the pBGCP. Transformed cells were cultured on kanamycin-free medium containing 2,4-D and carbenicillin for 2–3 weeks and then on the kanamycin medium for 3–4 months. The developed somatic embryos were transferred to the medium containing NAA, BA and kanamycin and subsequently regenerated into normal-appearing plants. Presence of the PRSV CP gene in the putative transgenic lines was detected by PCR and the expression of the CP was verified by Western blotting. The transgene was nuclearly inherited as revealed by segregation analysis in the backcrossed R1 progeny. From five independent experiments, the average successful rate of transformation was 15.9% of the zygotic embryos treated (52 transgenic somatic embryo clusters out of 327 zygotic embryos treated), about 10–100 times higher than the available methods previously reported. Thus, wounding highly regenerable differentiating tissues by carborundum vortexing provides a simple and efficient way for papaya transformation mediated byAgrobacterium.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chen MH, Chen CC (1992) Plant Cell Rep 11:404–407
Chen MH, Wang PJ, Maeda E (1987) Plant Cell Rep 6:348–351
DeBruijne E, DeLanghe E, van Rijck R (1974) Int Symp Fytofarm Fytiata 26:637–645
Fitch MMM, Manshardt RM (1990) Plant Cell Rep 9:320–324
Fitch MMM, Manshardt RM, Gonsalves D, Slightom JL (1993) Plant Cell Rep 12:245–249
Fitch MMM, Manshardt RM, Gonsalves D, Slightom JL, Sanford JC (1990) Plant Cell Rep 9:189–194
Fromm ME, Taylor LP, Walbot V (1986) Nature 319:791–793
Fitchen JH, Beachy R (1993) Annu Rev Microbial 47:739–763
Klee H, Horsch R, Rogers S (1987) Annu Rev Plant Physiol 38:467–486
Klein TM, Wolf ED, Wu R, Sanford JC (1987) Nature 327:70–73
Krens FA, Molendijk L, Wullems GJ, Schilperoort RA (1982) Nature 296:72–74
Laemmli UK (1970) Nature 227:680–685
Litz RE, Conover RA (1982) Plant Sci Lett 26:153–158
Litz RE, O'Hair SK, Conover RA (1983) Scientia Hort 19:287
Mettler IJ (1987) Plant Mol Biol Rep 5:346–349
Murashige T, Skoog F (1962) Physiol Plant 15:472–497
Pang SZ, Sanford JC (1988) J Amer Soc Hort Sci 113:287–291
Rogers SG, Horsch RB, Fraley RT (1986) Methods Enzymol 118:627–640
Tsay HS, Su CY (1985) Plant Cell Rep 4:28–30
Wang CH, Bau HJ, Yeh SD (1994) Phytopathology 84, 1205–1210
Yang JS, Ye CA (1992) Bot Bull Acad Sin 33:377–382
Yang JS, Yu TA, Cheng YH, Yeh SD (1996) Plant Cell Rep (in press)
Yeh SD, Gonsalves D, Provvidenti R (1984) Phytopathology 74, 1081–1085
Yeh SD, Jan FJ, Chiang CH, Doong TJ, Chen MC, Chung PH, Bau HJ (1992) J Gen Virol: 2531–2541
Yie ST, Liaw SI (1977) In Vitro 13:564–567
Author information
Authors and Affiliations
Additional information
Communicated by A. Komamine
Rights and permissions
About this article
Cite this article
Cheng, YH., Yang, JS. & Yeh, SD. Efficient transformation of papaya by coat protein gene of papaya ringspot virus mediated byAgrobacterium following liquid-phase wounding of embryogenic tissues with caborundum. Plant Cell Reports 16, 127–132 (1996). https://doi.org/10.1007/BF01890852
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01890852