Abstract
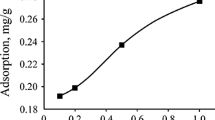
The waste slurry generated in fertilizer plants in India has been converted into a cheap carbonaceous adsorbent material. The prepared adsorbent has been characterised and used for the removal of lead and chromium metals. The kinetics of adsorption and the extent of adsorption at equilibrium are dependent on the physical and chemical characteristics of the adsorbate, adsorbent and experimental system. Results of laboratory scale studies conducted to delineate the effect of such parameters on the kinetics of adsorption of metal ions are reported. Parameters evaluated include: hydronium ion concentration, temperature, initial adsorbate concentration, size of adsorbent, and amount of adsorbent. On the basis of these studies the various physical parameters such as effective diffusion coefficient, activation energies and entropy of activation are evaluated, as these provide some information regarding the mechanistic aspects. Mass transfer coefficient values suggest a rapid transport of the adsorbate from bulk to solid phase.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
B.E. Reed and S. Arunachalam, Use of granular activated carbon columns for lead removal, Journal of Environ. Engg. Div., ASCE 120 (1994)416.
C.H. Weng and C.P. Huang, Treatment of metal industrial wastewater by fly ash and cement fixation. J. Environ. Engg. Div., ASCE 120 (1994) 1470.
K.K. Panday, G. Prasad and V.N. Singh, Mixed adsorbents for Cu(II) removal from aqueous solutions, Environ. Technol. Letters 7 (1986) 547–754.
C.K. Lee and K.S. Low, Removal of copper from solution using moss, Environ. Technol. Letters 10 (1989) 395–404.
Y.S. Ho, D.A.J. Waste and C.F. Forster, Batch nickel removal from aqueous solution by sphagnum moss peat, Wat. Res. 29 (1995) 1327–1332.
C.R. Nagendra Rao, L. Iyengar and C. Venkobachar, Sorption of copper(II) from aqueous phase by waste biomass, J. Environ. Engg. Div., ASCE 119 (1993) 369–377.
S. Kesraoul-Oukl, C. Cheeseman and R. Perry, Effect of conditioning and treatment of chabazite and clinoptilolite prior to lead and cadmium removal, Environ. Sci. Technol. 27 (1993) 1108–1116.
A. Groffman, S. Peterson and D. Brookins, Removing lead from wastewater using zeolites, Waster Environ. Technol. 5 (1992) 54.
D.P. Rodda, B.B. Johnson and J.D. Wells, The effect of temperature and pH on the adsorption of copper(II), lead(II) and zinc(II) onto goethite, J. Colloid Interface Science 161 (1993) 57–62.
D. Koeppenkastrop and H.D. Carlo Eric, Uptake of rare earth elements from solution by metal oxides, Environ. Sci. Technol., 27 (1993) 1796–1802.
S.K. Srivastava, G.B. Bhattacharjee, R. Tyagi, N. Pant and N. Pal, Studies on the removal of some toxic metal ions form aqueous solutions and industrial waste. Part 1: Removal of lead and cadmium by hydrous iron and aluminium oxide. Environ. Technol. Letters 99 (1988) 1173–1185.
S.K. Srivastava, A.K. Singh and A. Sharma, Studies on the uptake of lead and zinc by lignin from black liquior—a paper industry waste material, Environ. Technol. 15 (1994) 353–361.
S.K. Srivastava, R. Tyagi, N. Pant and N. Pal, Studies on the removal of some toxic metal ions. Part II: Removal of lead and cadmium by montmorillonite and kaolinite. Environ. Letters 10 (1989) 275–282.
A.K. Singh, D.P. Singh and V.N. Singh, Removal of Zn(II) by adsorption on China clay, Environ. Technol. Letters 9 (1988) 1153–1162.
T.W. Tee and R. Khan Majid, Removal of lead, cadmium and zinc by waste tea leaves. Environ. Technol. Letters 9 (1988) 1223.
S.J.T. Pollard, G.D. Fowler, C.J. Sollars and R. Perry, Low cost adsobents for waste and wastewater treatment: a review, The Science of the Total Environ. 116 (1992) 31–51.
S.K. Srivastava and N. Pant and N. Pal, Studies on the efficiency of a local fertilizer waste as a low cost adsorbent, Wat. Res. 11 (1987) 1389–1394.
S.K. Srivastava and R. Tyagi, Removal of substituted phenols by activated carbon (developed from fertilizer waste) and some thermodynamic aspects. Fresenius Envion. Bull. 3 (1994) 12–17.
S.K. Srivastava, R. Tyagi and N. Pant, Adsorption of heavy metal ions on carbonaceous material developed from the waste slurry generated in local fertilizer plants, Water Res. 23 (1989) 1161–1165.
J.S. Mattson and H.B. Mark,Activated Carbon Surface Chemistry and Adsorption from Aqueous Solution, Marcel Dekker, New York, 1971.
H.A. Szymanski and N.L. Alpert,Theory and Practice of Infrared Spectroscopy, Plenum, New York 1964.
F. Helfferich,Ion Exchange, McGraw-Hill, New York
C.E. Boyd, A.W. Adamson and L.S. Meyers, The exchange adsorption of ions from aqueous solution by organic zeolites II, Kinetics, J. Am. Chem. Soc. 59 (1947) 2836.
D. Reichenberg, Properties of ion-exchange resins in relation to their structure, III. Kinetics of exchange, J. Am. Chem. Soc. 75 (1953) 589.
J.P. Rawat and D.K. Singh, The kinetics of Ag, Zn, Cd, Hg, La and Th exchange in iron(III) antimonate, J. Inorg. Nucl. Chem. 40 (1978) 897.
J.P. Rawat and P.S. Thind, A kinetic study of ion-exchange in fontalum arsenate to understand the theoretical aspects of a separation, J. Phys. Chem. 80 (1976) 1384.
D.C. Freeman Jr. and D.N. Stamires, Electrical conductivity of synthetic crystalline zeolites, J. Chem. Phys. 35 (1961) 799.
G. Mckay, J.S. Allen, I.L. McConvey and M.S. Otterburn, Transport processes in the sorption of colored ions by peat particles, J. Colloid Int. Sci. 20 (1981) 323.
K. Periasamy and C. Namasivayam, Process development for removal and recovery of cadmium from wastewater by a low cost adsorbent: adsorption rates and equilibrium studies, Ind. Engg. Chem. Res. 33 (1994) 317.
C.F. Forster and D.C. Sharma, Removal of hexavalent chromium using sphagnum moss peat, Wat. Res. 27 (1993) 1201.
K.S. Low and C.K. Lee, Cadmium uptake by the mosscalymeves delessertii Besch, Bioresource Technol. 38 (1991) 1.
T.C. Tan, C.K. Chia and C.K. Teo, Uptake of metal ions by chemically treated human hair, Wat. Res. 88 (1992) 2227.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Srivastava, S.K., Gupta, V.K. & Mohan, D. Kinetic parameters for the removal of lead and chromium from wastewater using activated carbon developed from fertilizer waste material. Environ Model Assess 1, 281–290 (1996). https://doi.org/10.1007/BF01872156
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01872156