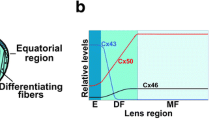
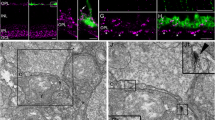
Summary
Lens epithelial cells are physiologically coupled to each other and to the lens fibers by an extensive network of intercellular gap junctions. In the rat, the epithelial-epithelial junctions appear to contain connexin43, a member of the connexin family of gap junction proteins. Limitations on the use of rodent lenses for the study of gap junction formation and regulation led us to examine the expression of connexin43 in embryonic chick lenses. We report here that chick connexin43 is remarkably similar to its rat counterpart in primary amino acid sequence and in several key structural features as deduced by molecular cDNA cloning. The cross-reactivity of an anti-rat connexin43 serum with chick connexin43 permitted definitive immunocytochemical localization of chick connexin43 to lens epithelial gap junctional plaques and examination of the biosynthesis of connexin43 by metabolic radiolabeling and immunoprecipitation. We show that chick lens cells synthesize connexin43 as a single, 42-kD species that is efficiently posttranslationally converted to a 45-kD form. Metabolic labeling of connexin43 with32P-orthophosphate combined with dephosphorylation experiments reveals that this shift in apparent molecular weight is due solely to phosphorylation. These results indicate that embryonic chick lens is an appropriate system for the study of connexin43 biosynthesis and demonstrate for the first time that connexin43 is a phosphoprotein.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Atkinson, M.M., Menko, A.S., Johnson, R.G., Sheppard, J.R., Sheridan, J.D. 1981. Rapid and reversible reduction of junctional permeability in cells infected with a temperature-sensitive mutant of avian sarcoma virus.J. Cell Biol. 91:573–578
Azarnia, R., Reddy, S., Kmiecik, T.C., Shalloway, D., Loewenstein, W.R. 1988. The cellularsrc gene product regulates junctional cell-to-cell communication.Science 239:398–401
Benedetti, E.L., Dunia, I., Bloemendal, H. 1974. Development of junctions during differentiation of lens fibers.Proc. Natl. Acad. Sci. USA 71:5073–5077
Beyer, E.C., Goodenough, D.A., Paul, D.L. 1988. The connexins, a family of related gap junction proteins.In: Gap Junctions. E.L. Hertzberg and R.G. Johnson, editors. pp. 167–175. Alan R. Liss, New York
Beyer, E.C., Kistler, J., Paul, D.L., Goodenough, D.A. 1989. Antisera directed against connexin43 peptides react with a 43-kD protein localized to gap junctions in myocardium and other tissues.J. Cell Biol. 108:595–605
Beyer, E.C., Paul, D.L., Goodenough, D.A. 1987. Connexin43: A protein from rat heart homologous to a gap junction protein from liver.J. Cell Biol. 105:2621–2629
Burt, J.M., Spray, D.C. 1988a. Inotropic agents modulate gap junctional conductance between cardiac myocytes.Am. J. Physiol. 254:H1206-H1210
Burt, J.M., Spray, D.C. 1988b. Single-channel events and gating behavior of the cardiac gap junction channel.Proc. Natl. Acad. Sci. USA 85:3431–3434
Chirgwin, J.M., Przybyla, A.E., MacDonald, R.J., Rutter, W.J. 1979. Isolation of biologically active ribonucleic acid from sources enriched in ribonnuclease.Biochemistry 18:5294–5299
Ebihara, L., Beyer, E.C., Swenson, K.I., Paul, D.L., Goodenough, D.A. 1989. Cloning and expression of aXenopus embryonic gap juction protein.Science 243:1194–1195
Feinberg, A.P., Vogelstein, B. 1983. A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity.Anal. Biochem. 132:6–13
FitzGerald, P.G., Goodenough, D.A. 1986. Rat lens cultures: MIP expression and domains of intercellular coupling.Invest. Ophthalmol. Vis. Sci. 27:755–771
Flagg-Newton, J.L., Dahl, G., Loewenstein, W.R. 1981. Cell junction and cyclic AMP: I. Upregulation of junctional membrane permeability and junctional membrane particles by administration of cyclic nucleotide or phosphodiesterase inhibitor.J. Membrane Biol. 63:105–121
Gimlich, R.L., Kumar, N.M., Gilula, N.B. 1988. Sequence and developmental expression of mRNA coding for a gap junction protein inXenopus.J. Cell Biol. 107:1065–1073
Goodenough, D.A., Dick, J.S.B., II, Lyons, J.E. 1980. Lens metabolic cooperation: A study of mouse lens transport and permeability visualized with freeze-substitution autoradiography and electron microscopy.J. Cell Biol. 86:576–589
Goodenough, D.A., Paul, D.L., Jesaitis, L. 1988. Topological distribution of two connexin32 antigenic sites in intact and split rodent hepatocyte gap junctions.J. Cell Biol. 107:1817–1824
Goodenough, D.A., Revel, J.P. 1971. The permeability of isolated and in situ mouse hepatic gap junctions studied with enzymatic tracers.J. Cell Biol. 50:81–91
Gruijters, W.T.M., Kistler, J., Bullivant, S., Goodenough, D.A. 1987. Immunolocalization of MP70 in lens fiber 16–17nm intercellular junctions.J. Cell Biol. 104:565–572
Helms, C., Graham, M.Y., Dutchik, J.E., Olson, M.V. 1985. A new method for purifying Lambda DNA from phage lysates.DNA 4:39–49
Kistler, J., Christie, D., Bullivant, S. 1988. Homologies between gap junction proteins in lens, heart and liver.Nature (London) 331:721–723
Kistler, J., Kirkland, B., Bullivant, S. 1985. Identification of a 70,000-D protein in lens membrane junctional domains.J. Cell Biol. 101:28–35
Kumar, N.M., Gilula, N.B. 1986. Cloning and characterization of human and rat liver cDNAs coding for a gap junction protein.J. Cell Biol. 103:767–776
Kyte, J., Doolittle, R.F. 1982. A simple method for displaying the hydropathic character of a protein.J. Mol. Biol. 157:105–132
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4.Nature (London) 227:680–685
Loewenstein, W.R. 1985. Regulation of cell-to-cell communication by phosphorylation.Biochem. Soc. Symp. London 50:43–58
Mathias, R.T., Rae, J.L. 1985. Transport properties of the lens.Am. J. Physiol. 249:C181-C190
McLean, I.W., Nakane, P.K. 1974. Periodate-lysine-paraformaldehyde fixative. A new fixative for immunoelectron microscopy.J. Histochem. Cytochem. 22:1077–1083
Meijer, L., Arion, D., Golsteyn, R., Pines, J., Brizuela, L., Hunt, T., Beach, D. 1989. Cyclin is a component of the sea urchin egg M-phase specific histone H1 kinase.EMBO J. 8:2275–2282
Melton, D.A., Krieg, P.A., Rebagliatti, M.R., Maniatis, T., Zinn, K., Green, M.R. 1984. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promotor.Nucleic Acids Res. 12:7035–7056
Menko, A.S., Klukas, K.A., Johnson, R.G. 1984. Chicken embryo lens cultures mimic differentiation in the lens.Dev. Biol. 103:129–141
Menko, A.S., Klukas, K.A., Liu, T.-F., Quade, B., Sas, D.F., Preus, D.M., Johnson, R.G. 1987. Junctions between lens cells in differentiating cultures: Structure, formation, intercellular permeability, and junctional protein expression.Dev. Biol. 123:307–320
Miller, T.M., Goodenough, D.A. 1985. Gap junction structures after experimental alteration of junctional channel conductance.J. Cell Biol. 101:1741–1748
Miller, T.M., Goodenough, D.A. 1986. Evidence for two physiologically distinct gap junctions expressed by the chick lens epithelial cell.J. Cell Biol. 102:194–199
Moria, A.O., Draetta, G., Beach, D., Wang, J.Y.J. 1989. Reversable tyrosine phosphorylation of cdc2: Dephosporylation accompanies activation during entry into mitosis.Cell 58:193–203
Musil, L.S., Carr, C., Cohen, J.B., Merlie, J.P. 1988. Acetylcholine receptor associated 43K protein contains covalently bound myristate.J. Cell Biol. 107:1113–1121
Nicholson, B.J., Zhang, J. 1988. Multiple protein components in a single gap junction: Cloning of a second hepatic gap junction protein (Mr 21,000).In: Gap Junctions. E.L. Hertzberg and R.G. Johnson, editors. pp. 207–218. Alan R. Liss, New York
Patchinsky, T., Hunter, T., Esch, F.S., Cooper, J.A., Sefton, B.M. 1982. Analysis of the sequence of amino acids surrounding sites of tyrosine phosphorylation.Proc. Natl. Acad. Sci. USA 79:973–977
Paul, D.L. 1986. Molecular cloning of cDNA for rat liver gap junction protein.J. Cell Biol. 103:123–134
Paul, D.L., Goodenough, D.A. 1983. Preparation, characterization, and localization of antisera against bovine MP26, an integral protein from lens fiber plasma membrane.J. Cell Biol. 96:625–632
Peracchia, C. 1978. Calcium effects on gap junction structure and cell coupling.Nature London 271:669–671
Rae, J.L., Kuszak, J.R. 1983. The electrical coupling of epithelium and fibers in the frog lens.Exp. Eye Res. 36:317–326
Ralston, R., Bishop, J.M. 1985. The product of the protooncogenec-src is modified during the cellular response to plateletderived growth factor.Proc. Natl. Acad. Sci USA 82:7845–7849
Saez, J.C., Spray, D.C., Nairn, A.C., Hertzberg, E., Greengard, P., Bennett, M.V.L. 1986. cAMP increases junctional conductance and stimulates phosphorylation of the 27-kDa principal gap junction polypeptide.Proc. Natl. Acad. Sci USA 83:2473–2477
Sanger, F., Nicklen, S., Coulsen, A.R. 1977. DNA sequencing with chain terminating inhibitors.Proc. Natl. Acad. Sci. USA 79:441–445
Schuetze, S.M., Goodenough, D.A. 1982. Dye transfer between cells of the embryonic chick lens becomes less sensitive to CO2-treatment with development.J. Cell Biol. 92:694–705
Swenson, K.I., Jordan, J.R., Beyer, E.C., Paul, D.L. 1989. Formation of gap junctions by expression of connexins inXenopus oocyte pairs.Cell 57:145–155
Traub, O., Look, J., Paul, D., Willecke, K. 1987. Cyclic adenosine monophosphate stimulates biosynthesis and phosphorylation of the 26kDa gap junction protein in cultured mouse hepatocytes.Eur. J. Cell Biol. 43:48–54
Wiener, E.C., Loewenstein, W.R. 1983. Correction of cell-cell communication defect by introduction of a protein kinase into mutant cells.Nature (London) 305:433–435
Zampighi, G., Hall, J.E., Ehring, G.R., Simon, S.A. 1989. The structural organization and protein composition of lens fiber junctions.J. Cell Biol. 108:2255–2275
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Musil, L.S., Beyer, E.C. & Goodenough, D.A. Expression of the gap junction protein connexin43 in embryonic chick lens: Molecular cloning, ultrastructural localization, and post-translational phosphorylation. J. Membrain Biol. 116, 163–175 (1990). https://doi.org/10.1007/BF01868674
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01868674