Summary
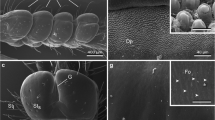
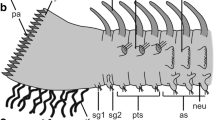
The ventral surface ofHolothuria forskali (Holothuroida, Aspidochirotida) is almost completely covered by small-sized podia that are locomotory. Each podium consists of a stem that allows the podium to lengthen, to flex, and to retract, and this is topped by a disc that allows the podium to adhere to the substratum during locomotion. Podia ofH. forskali do not end in a sucker and their adhesion to the substratum thus relies entirely on the disc epidermal secretions. The disc epidermis is made of five cell types: non-ciliated secretory cells of two different types that contain granules whose content is either mucopolysaccharidic (NCS1 cells) or mucopolysaccharidic and proteinic in nature (NCS2 cells), ciliated secretory cells containing small granules of unknown nature (CS cells), cilitated nonsecretory cells (CNS cells), and support cells. The cilia ofCS cells are subcuticular whereas those ofCNS cells, although also short and rigid, traverse the cuticle and protrude in the outer medium. During locomotion, epidermal cells of the podial disc are presumably involved in an adhesive/de-adhesive process functioning as a duogland adhesive system. Adhesive secretions would be produced byNCS1 andNCS2 cells and de-adhesive secretion byCS cells. All these secretions would be controlled by stimulations of the two types of ciliated cells (receptor cells) which presumably interact with the secretory cells by way of the nerve plexus. The lack of suckers and the coexistence of two adhesive cell types in the disc epidermis give the locomotory podia ofH. forskali a “compromise” structure which would perhaps explain their ability to move as efficiently along soft and hard substrata.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ball B, Jangoux M (1990) Ultrastructure of the tube foot sensorysecretory complex inOphiocomina nigra (Echinodermata, Ophiuridea). Zoomorphology 109:201–209
Bouland C, Massin C, Jangoux M (1982) The fine structure of the buccal tentacles ofHolothuria for skali (Echinodermata, Holothuroidea). Zoomorphology 101:133–149
Cameron JL, Fankboner PV (1984) Tentacle structure and feeding processes in life stages of the commercial sea cucumberParastichopus californicus (Stimpson). J Exp Mar Biol Ecol 81:193–209
Coleman R (1969) Ultrastructure of the tube foot sucker of a regular echinoid,Diadema antillarum Philippi, with special reference to the secretory cells. Z Zellforsch Mikrosk Anat 96:151–161
Dietrich HF, Fontaine AR (1975) A decalcification method for ultrastructure of echinoderm tissues. Stain Technol 50(5):351–354
Flammang P, De Ridder C, Jangoux M (1990) Morphologie fonctionnelle des podia pénicillés chez l'échinide fouisseurEchinocardium cordatum (Echinodermata). In: De Ridder C, Dubois Ph, Lahaye M-C, Jangoux M (eds) Echinoderm Research. Balkema, Rotterdam, pp 233–238
Flammang P, De Ridder C, Jangoux M (1991) Ultrastructure of the penicillate podia of the spatangoid echinoidEchinocardium cordatum (Echinodermata) with special emphasis on the epidermal sensory-secretory complexes. Acta Zool (Stockholm) 72:151–158
Florey E, Cahill MA (1977) Ultrastructure of sea urchin tube feet. Evidence for connective tissue involvement in motor control. Cell Tissue Res 177:195–214
Ganter P, Jollés G (1969–1970) Histochimie normale et pathologique, vol1 et 2. Gauthiers-Villars, Paris
Harrison G (1968) Subcellular particles in echinoderm tube feet. II. Class Holothuroidea. J Ultrastruct Res 23:124–133
Harrison G, Philpott D (1966) Subcellular particles in echinoderm tube feet. I. Class Asteroidea. J Ultrastruct Res 16:537–547
Hermans CO (1983) The duo-gland adhesive system. Oceanogr Mar Biol Ann Rev 21:281–339
Hyman LH (1955) The Invertebrates, vol 4. Echinodermata. McGraw-Hill, New York
Kawaguti S (1964) Electron microscopic structure of the podial wall of an echinoid with special references to the nerve plexus and the muscle. Biol J Okayama Univ 10:1–12
Lahaye M-C, Jangoux M (1985) Functional morphology of the podia and ambulacral grooves of the comatulid crinoidAntedon bifida (Echinodermata). Mar Biol 86:307–318
Lawrence JM (1987) A functional Biology of Echinoderms. Croom Helm, London, Sidney
McKenzie JD (1987) The ultrastructure of the tentacles of eleven species of dendrochirote holothurians studied with special reference to surface coats and papillae. Cell Tissue Res 248:187–199
McKenzie JD (1988a) Ultrastructure of the tentacles of the apodous holothurianLeptosynapta spp (Holothuroidea:Echinodermata) with special reference to the epidermis and surface coats. Cell Tissue Res 251:387–397
McKenzie JD (1988b) The ultrastructure of tube foot epidermal cells and secretions: their relationship to the duo-glandular hypothesis and the phylogeny of the echinoderm classes. In: Paul CRC, Smith AB (eds) Echinoderm Phylogeny and Evolutionary Biology. Clarendon Press, Oxford, pp 287–298
Menton DN, Eisen AZ (1970) The structure of the integument of the sea cucumber,Thyone briareus. J Morphol 131:17–36
Nichols D (1966) Functional morphology of the water-vascular system. In: Boolootian RA (ed) Physiology of Echinodermata. Interscience Publishers, New York, pp 219–240
Sedmak JJ, Grossberg SE (1977) A rapide, sensitive, and versatile assay for protein using Coomassie Brilliant blue G250. Anal Biochem 79:544–552
Smith JE (1937) The structure and function of the tube feet in certain echinoderms. J Mar Biol Assoc UK 22:345–357
Smith TB (1983) Tentacular ultrastructure and feeding behaviour ofNeopentadactyla mixta (Holothuroidea:Dendrochirota). J Mar Biol Assoc UK 63:301–311
Souza Santos H, Silva Sasso W (1968) Morphological and histochemical studies on the secretory glands of starfish tube feet. Acta Anat 69:41–51
Wood RL, Cavey MJ (1981) Ultrastructure of the coelomic lining in the podium of the starfishStylasterias forreri. Cell Tissue Res 218:449–473
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Flammang, P., Jangoux, M. Functional morphology of the locomotory podia ofHolothuria forskali (Echinodermata, Holothuroida). Zoomorphology 111, 167–178 (1992). https://doi.org/10.1007/BF01632906
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01632906