Summary
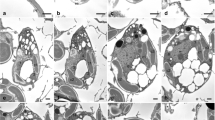
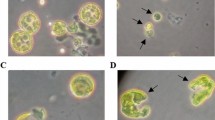
Each wild-typeChlamydomonas reinhardtii cell has one large chloroplast containing several nuclei (nucleoids). We used DNA insertional mutagenesis to isolate Chlamydomonas mutants which contain a single, large chloroplast (cp) nucleus and which we namedmoc (monokaryotic chloroplast). DAPI-fluorescence microscopy and microphotometry observations revealed thatmoc mutant cells only contain one cp-nucleus throughout the cell division cycle, and that unequal segregation of cpDNA occurred during cell division in themoc mutant. One cell with a large amount of cpDNA and another with a small amount of cpDNA were produced after the first cell division. Unequal segregation also occurred in the second cell division, producing one cell with a large amount (about 70 copies) of cpDNA and three other cells with a small amount (only 2–8 copies) of cpDNA. However, most individualmoc cells contained several dozen cpDNA copies 12 h after the completion of cell division, suggesting that cpDNA synthesis was activated immediately after chloroplast division. In contrast to the cpDNA, the mitochondrial (mt) DNA of themoc mutants was observed as tiny granules scattered throughout the entire cell. These segregated to each daughter cell equally during cell division. Electron-microscopic observation of the ultrastructure ofmoc mutants showed that a low-electron-density area, which was identified as the cp-nucleus by immunoelectron microscopy with anti-DNA antibody, existed near the pyrenoid. However, there were no other structural differences between the chloroplasts of wild-type cells andmoc mutants. The thylakoid membranes and pyrenoid were identical. Therefore, we propose that the novelmoc mutants are only defective in the dispersion and segregation of cpDNA. This strain should be useful to elucidate the mechanism for the segregation of cpDNA.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- DAPI:
-
4′,6-diamidino-2-phenylindole
- VIMPCS:
-
video-intensified microscope photon-counting system
References
Ehara T, Ogasawara Y, Osafune T, Hase E (1990) Behavior of chloroplast nucleoids during the cell cycle ofChlamydomonas reinhardtii (Chlorophyta) in synchronized culture. J Phycol 26: 317–323
Fernandez E, Schnell R, Ranum LPW, Hussey SC, Silflow CD, Lefebvre PA (1989) Isolation and characterization of the nitrate reductase structural gene ofChlamydomonas reinhardtii. Proc Natl Acad Sci USA 86: 6449–6453
Goodenough UW (1970) Chloroplast division and pyrenoid formation inChlamydomonas reinhardtii. J Phycol 6: 1–6
Gumpel NJ, Purton S (1994) Playing tag withChlamydomonas. Trends Cell Biol 4: 299–301
Hansmann P, Falk H, Ronai K, Sitte P (1985) Structure, composition, and distribution of plastid nucleoids inNarcissus pseudonarcissus. Planta 164: 459–472
Harris EH (1989) TheChlamydomonas sourcebook: a comprehensive guide to biology and laboratory use. Academic Press, San Diego
Hashimoto H (1985) Changes in the distribution of nucleoids in developing and dividing chloroplasts and etioplasts ofAvena sativa. Protoplasma 127: 119–127
Itoh R, Takahashi H, Toda K, Kuroiwa H, Kuroiwa T (1997) DNA gyrase involvement in chloroplast-nucleoid division inCyanidioschyzon merolae. Eur J Cell Biol 73: 252–258
Kindle KL (1990) High-frequency nuclear transformation ofChlamydomonas reinhardtii. Proc Natl Acad Sci USA 87: 1228–1232
—, Schnell RA, Fernandez E, Lefebvre PA (1989) Stable nuclear transformation ofChlamydomonas using theChlamydomonas gene for nitrate reductase. J Cell Biol 109: 2589–2601
Kuroiwa T (1991) The replication, differentiation, and inheritance of plastids with emphasis on the concept of organelle nuclei. Int Rev Cytol 128: 1–62
—, Suzuki T, Ogawa K, Kawano S (1981) The chloroplast nucleus: distribution, number, size, and shape and a model for the multiplication of the chloroplast genome during chloroplast development. Plant Cell Physiol 22: 381–396
—, Kuroiwa H, Sakai A, Takahashi H, Toda K, Itoh R (1998) The division apparatus of plastid and mitochondria. Int Rev Cytol 181: 1–41
Lindbeck AGC, Rose RJ (1987) The role of chloroplast membranes in the location of chloroplast DNA during the greening ofPhaseolus vulgaris etioplasts. Protoplasma 139: 92–99
Liu JW, Rose RJ (1992) The spinach chloroplast chromosome is bound to the thylakoid membrane in the region of the inverted repeat. Biochem Biophys Res Commun 184: 993–1000
Maguire MJ, Goff LJ, Coleman AW (1995) In situ plastid and mitochondrial DNA determination: implication of the observed minimal genome number. Am J Bot 82: 1496–1506
Marisson JL, Leech RM (1992) Co-immunolocalization of topoisomerase II and chloroplast DNA in developing, dividing and mature wheat chloroplasts. Plant J 2: 783–790
Mihara S, Hase E (1971) Studies on the vegetative life cycle ofChlamydomonas reinhardti Dangeard in synchronous culture I: some characteristics of the cell cycle. Plant Cell Physiol 12: 225–236
Miyamura S, Nagata T, Kuroiwa T (1986) Quantitative fluorescence microscopy on dynamic changes of plastid nucleoids during wheat development. Protoplasma 133: 66–72
Nakamura S, Itoh S, Kuroiwa T (1986) Behavior of chloroplast nucleus during chloroplast development and degeneration inChlamydomonas reinhardtii. Plant Cell Physiol 27: 775–784
Nemoto Y, Nagata T, Kuroiwa T (1989) Studies of plastid-nuclei (nucleoids) inNicotiana tabacum L. II: disassembly and reassembly of proplastid-nuclei isolated from culture cells. Plant Cell Physiol 30: 445–454
—, Kawano S, Kondoh K, Nagata T, Kuroiwa T (1990) Studies of plastid-nuclei (nucleoids) inNicotiana tabacum L. III: isolation of chloroplast-nuclei from mesophyll protoplasts and identification of chloroplast DNA-binding proteins. Plant Cell Physiol 31: 767–776
Nishimura, Y, Higashiyama T, Suzuki L, Misumi O, Kuroiwa T (1998) The biparental transmission of the mitochondrial genome inChlamydomonas reinhardtii visualized in living cells. Eur J Cell Biol 77: 124–133
Rose RJ (1988) The role of membranes in the segregation of plastid DNA. In: Boffey SA, Lloyd D (eds) Division and segregation of organelles. Cambridge University Press, Cambridge, pp 171–195 (Society of Experimental Biologists seminar series, vol 35)
Sager R, Granick S (1953) Nutritional studies inChlamydomonas reinhardtii. Ann N Y Acad Sci 56: 831–838
Sato N, Albrieux C, Joyaed J, Douce R, Kuroiwa T (1993) Detection and characterization of a plastid envelope DNA-binding protein which may anchor plastid nucleoid. EMBO J 12: 555–561
—, Misumi O, Shinada Y, Sasaki M, Yoine M (1997) Dynamics of localization and protein composition of plastid nucleoids in light-grown pea seedlings. Protoplasma 200: 163–173
—, Ohshima K, Watanabe A, Ohta N, Nishiyama Y, Joyaed J, Douce R (1998) Molecular characterization of the PEND protein, a novel bZIP protein present in the envelope membrane that is the site of nucleoid replication in developing plastids. Plant Cell 10: 859–972
Sodmergen, Kawano S, Tano S, Kuroiwa T (1991) Degradation of chloroplast DNA in second leaves of rice (Oryza sativa) before leaf yellowing. Protoplasma 160: 89–98
Stevens DR, Purton S (1997) Genetic engineering of eukaryotic algae: progress and prospects. J Phycol 33: 713–722
Tam LW, Lefebvre PA (1993) Cloning flagellar genes inChlamydomonas reinhardtii by DNA insertional mutagenesis. Genetics 135: 375–384
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Misumi, O., Suzuki, L., Nishimura, Y. et al. Isolation and phenotypic characterization ofChlamydomonas reinhardtii mutants defective in chloroplast DNA segregation. Protoplasma 209, 273–282 (1999). https://doi.org/10.1007/BF01453455
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01453455