Abstract
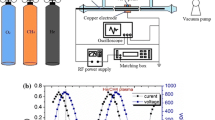
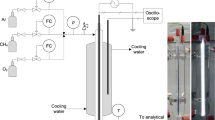
Conversion of CH4 with a N2 microwave plasma (2.45 GHz) is studied. The experiments cover the absorbed microwave power range 300–700 W with 17–62% of methane in the gas mixture, with pressures of 10–40 mbar and flow rates of 140–650 ml· min−1. The yields of C2 hydrocarbons and dihydrogen are analyzed by gas chromatography. The distance of methane addition downstream of the plasma plays an important role on the composition and the concentration of the products obtained. This distance mainly determines the energy concentrated in the active species of the plasma when they react with methane. Different behaviors for acetylene formation, on the one hand, and for ethane and ethene formation, on the other hand, have been observed, and this finding allows us to propose a kinetic mechanism for the decay of methane and for the formation of C2 hydrocarbons.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
F. Raulin, D. Mourey, and G. Toupance,Origins Life 12, 267 (1982).
W. R. Thomson, T. J. Henry, J. M. Schwartz, B. N. Khare, and C. Sagan,Icarus 90, 57 (1991).
M. E. Fraser, D. A. Fee, and R. S. Sheinson,Plasma Chem. Plasma Process. 5, 163 (1985).
D. E. Tevault,Plasma Chem. Plasma Process. 5, 369 (1985).
A. M. Diamy, J. C. Legrand, J. Al Andari, L. Hochard, and A. Ricard,11th ESCAMPIG, August 1992, St. Petersbourg (CEI), inEurophysics Conference Abstracts 16F, 203 (1992).
H. Michel,Interactions Plasmas Froids Materiaux, Les Editions de Physique, Les Ulis (1987), p. 465.
J. Amouroux and D. Rapakoulias,Rev. Phys. Appl. 12, 1013 (1977); D. Rapakoulias and J. Amouroux,ibid.,14, 961 (1979). P. Capezzuto, F. Cramarossa, G. Ferraro, P. Maione, and E. Molinari,Gazz. Chim. Ital. 103, 1153 (1973). P. Capezzuto, F. Cramarossa, R. D'Agostino, and E. Molinari,ibid.,103, 1169 (1973).
A. Oumghar, J. C. Legrand, A. M. Diamy, N. Turillon, and R. I. Ben-Aim,10th Int. Svmp. Plasma Chem- Bocluan (Germany), Proc. 3, 2.3–7 (1991).
J. Weill, F. Chevron, C. Raimbault, R. Genier, G. Renesme, L. Capogna, and Y. Muller,Rev. Inst. Fr. Pétrole 47, 255 (1992). J. H. Kiefer and S. S. Kumaran,J. Phys. Chem. 97, 414 (1993).
J. Al Andari, A. M. Diamy, V. Hrachová, J. C. Legrand, and N. Turillon,11th. Int. Symp. Plasma Chem., Loughborough (England), Proc. 4, 1344 (1993).
A. N. Wright and C. A. Winkler,Active Nitrogen, Academic Press, New York (1968).
P. Nectoux, R. Derai, and J. Danon,Chem. Phys. 37, 229 (1979).
W. L. Morgan,Plasma Chem. Plasma Process. 12, 477 (1992).
M. F. Golde,Int. J. Chem. Kinet. 20, 751 (1988).
M. F. Golde, G. H. Ho, W. Tao, and J. M. Thomas,J. Phys. Chem. 93, 1112 (1989).
A. N. Wright and C. A. Winkler,Can. J. Chem. 40, 1291 (1962).
S. P. Walch and R. J. Duchovic,J. Chem. Phys. 90, 3230 (1989).
M. Capitelli and E. Molinari,Top. Curr. Chem. 90, 59 (1980).
M. Capitelli, C. Gorse, and A. Ricard,Top. Curr. Phys. 39, 315 (1986).
A. Ricard,Interactions Plasmas Froids Matériaux, Les Editions de Physique, Les Ulis (1987), p. 241.
L. G. Piper,J. Chem. Phys. 87, 1625 (1987).
W. J. Marinelli, W. J. Kessler, B. D. Green, and W. A. M. Blumberg,J. Chem. Phys. 90, 2167 (1989).
D. L. Baulch, C. J. Cobos, R. A. Cox, C. Esser, P. Frank, T. Just, J. A. Kerr, M. J. Pilling, J. Troe, R. W. Walker, and J. Warnatz,J. Phys. Chem. Ref. Data 21, 411 (1992).
P. Glarborg, J. A. Miller, and R. J. Kee,Combust. Flame 65, 177 (1986).
F. L. Nesbitt, G. Marston, and L. J. Stief,J. Phys. Chem. 94, 4946 (1990).
I. Messing, S. V. Filseth, C. M. Sadowski, and T. Carrington,J. Chem. Phys. 74, 3874 (1981).
K. Schofield,J. Phys. Chem. Ref Data 8, 723 (1979).
B. Fell, I. V. Rivas, and D. L. McFadden,J. Phys. Chem. 85, 224 (1981).
H. Umemoto, K. Sugiyama, S. Tsunashima, and S. Sato,Bull. Chem. Soc. Jpn. 58, 3076 (1985).
M. Tichy, A. B. Rakshit, D. G. Lister, N. D. Twiddy, N. G. Adams, and D. Smith,Int. J. Mass Spectrom. Ion Process. 29, 231 (1979).
K. Hiraoka, K. Aoyama, and K. Morise,Can. J. Chem. 63, 2899 (1985).
J. Warnatz, Rate Coefficients in the C/H/O System, inCombustion Chemistry, W. C. Gardiner, Jr., ed., Springer-Verlag, New York (1984), Chap. 5, p. 197.
J. C. Mackie and K. R. Doolan,Int. J. Chem. Kinet. 16, 525 (1984).
W. Braun, K. H. Welge, and J. R. McNesby,J. Chem. Phys. 45, 2650 (1966).
K. S. Yin and M. Venugopalan,Plasma Chem. Plasma Process 3, 343 (1983), M. Touvelle, J. L. Munoz Licea, and M. Venugopalan,ibid. 7, 101 (1987).
H. Uyama and O. Matsumoto,Plasma Chem. Plasma Process. 9, 13, 421 (1989). H. Uyama, T. Nakamura, S. Tanaka, and O. Matsumoto,ibid.,13, 117 (1993).
W. Tsang and R. F. Hampson,J. Phys. Chem. Ref. Data 15, 1087 (1986).
W. Braun, J. R. McNesby, and A. M. Bass,J. Chem. Phys. 46, 2071 (1967).
L. Herbert, I. W. M. Smith, and R. D. Spencer-Smith,Int. J. Chem. Kinet. 24, 791 (1992).
A. J. Dean, R. K. Hanson, and C. T. Bowman,J. Phys. Chem. 95, 3180 (1991).
W. A. Sanders and M. C. Lin, Kinetics and Mechanisms of Methylidyne Radical Reactions, inChemical Kinetics of Small Organic Radicals, Z. B. Alfassi, ed., CRC Press, Boca Raton, Florida (1988), Vol. 3, p. 103.
C. P. Tsaï and D. L. McFadden,J. Phys. Chem. 94, 3298 (1990).
J. Al Andari, A. M. Diamy, J. C. Legrand, and R. I. Ben-Aim,Plasma Chem. Plasma Process. 13, 103 (1993).
M. W. Chase, C. A. Davies, J. R. Downey, D. J. Frurip, R. A. McDonald, and A. N. Syverud, JANAF Thermochemical Tables,J. Phys. Chem. Ref. Data 14, Suppl. No. 1, 1 (1985).
French Patent 92-01455 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oumghar, A., Legrand, J.C., Diamy, A.M. et al. A kinetic study of methane conversion by a dinitrogen microwave plasma. Plasma Chem Plasma Process 14, 229–249 (1994). https://doi.org/10.1007/BF01447080
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01447080