Summary
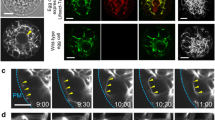
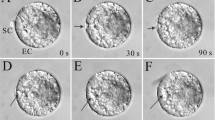
Actin organization was observed inm-maleimidobenzoic acid N-hydroxysuccinimide ester(MBS)-treated maize embryo sacs by confocal laser scanning microscopy. The results revealed that dynamic changes of actin occur not only in the degenerating synergid, but also in the egg during fertilization. The actin filaments distribute randomly in the chalazal part of the synergid before fertilization; they later become organized into numerous aggregates in the chalazal end after pollination. The accumulation of actin at this region is intensified after the pollen tube discharges its contents. Concurrently, actin patches have also been found in the cytoplasm of the egg cell and later they accumulate in the cortical region. To compare with MBS-treated maize embryo sacs, we have performed phalloidin microinjection to label the actin cytoskeleton in living embryo sacs ofTorenia fournieri. The results have extended the previous observations on the three-dimensional organization of the actin arrays in the cells of the female germ unit and confirm the occurrence of the actin coronas in the embryo sac during fertilization. We have found that there is an actin cap occurring near the filiform apparatus after anthesis. In addition, phalloidin microinjection into the Torenia embryo sac has proved the presence of intercellular actin between the cells of the female germ unit and thus confirms the occurrence of the actin coronas in the embryo sac during fertilization. Moreover, actin dynamic changes also take place in the egg and the central cell, accomplished with the interaction between the male and female gametes. The actin filaments initially organize into a distinct actin network in the cortex of the central cell after anthesis; they become fragmented in the micropylar end of the cell after pollination. Similar to maize, actin patches have also been observed in the egg cortex after pollination. This is the first report of actin dynamics in the living embryo sac. The results suggest that the actin cytoskeleton may play an essential role in the reception of the pollen tube, migration of the male gametes, and even gametic fusion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- DIC:
-
differential interference contrast
- MBS:
-
m-maleimidobenzoic acid N-hydroxysuccinimide ester
References
Bednara J, Willemse MTM, van Lammeren AAM (1990) Organization of the actin cytoskeleton during megasporogenesis inGasteria verrucosa visualized with fluorescent-labeled phalloidin. Acta Bot Neerl 39: 43–48
Chaubal R, Reger BJ (1990) Relatively high calcium is localized in synergid cells of wheat ovaries. Sex Plant Reprod 3: 98–102
— — (1992) The dynamics of calcium distribution in the synergid cells of wheat after pollination. Sex Plant Reprod 5: 206–213
Cleary AL (1995) F-actin redistribution at the division site in livingTradescantia stomatal complexes as revealed by microinjection of rhodamine-phalloidin. Protoplasma 185: 152–165
Doris FP, Steer MW (1996) Effects of fixatives and permeabilization buffer on pollen tubes: implications for localization of actin microfilaments using phalloidin staining. Protoplasma 195: 25–36
Grolig F (1998) Nuclear centering inSpirogyra: force integration by microfilaments along microtubules. Planta 204: 54–63
Hart NH, Fluck RA (1995) Cytoskeleton inToleost eggs and early embryo: contribution to cytoarchitecture and mobile events. Curr Top Dev Biol 31: 343–381
Heslop-Harrison J, Heslop-Harrison Y (1991) The actin cytoskeleton in unfixed pollen tubes following microwave-accelerated DMSO-permeabilization and TRITC-phalloidin staining. Sex Plant Reprod 4: 6–11
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (1997) Kinetics of double fertilization inTorenia fournieri based on direct observations of the naked embryo sac. Planta 203: 101–110
Huang BQ, Russell SD (1992) Synergid degeneration inNicotiana: a quantitative, fluorochromatic and chlorotetracycline study. Sex Plant Reprod 5: 151–155
— — (1994) Fertilization inNicotiana tabacum: cytoskeletal modifications in the embryo sac during synergid degeneration. Planta 194: 200–214
—, Sheridan FW (1994) Female gametophyte development in maize: microtubular organization and embryo sac polarity. Plant Cell 6: 845–861
— — (1998) Actin coronas in normal and indeterminate gametophyte embryo sacs of maize. Sex Plant Reprod 11: 257–264
—, Pierson ES, Russell SD, Tiezzi A, Cresti M (1992) Video microscopic observations of living, isolated embryo sacs ofNicotiana and their component cells. Sex Plant Reprod 5: 156–162
— — — — — (1993) Cytoskeletal organization and modification during pollen tube arrival, gamete delivery and fertilization inPlumbago zeylanica. Zygote 1: 143–154
Jensen WA (1974) Reproduction in flowering plants. In: Rodard AW (ed) Dynamic aspects of plants ultrastructure. McGraw-Hill, New York, pp 481–503
Keijzer CJ (1992) The isolation of sperm cells, their microinjection into the egg apparatus and method for structural analysis of the injected cells. In: Cresti M, Tiezzi A (eds) Sexual plant reproduction. Springer, Berlin Heidelberg New York Tokyo, pp 161–171
—, Reinders MC, Leferinkten Klooster HB (1988) A microinjection method for artificial fertilization inTorenia. In: Cresti M, Gori P, Pacini E (eds) Sexual reproduction in higher plants. Springer, Berlin Heidelberg New York Tokyo, pp 119–124
Lloyd CW (1989) The plant cytoskeleton. Curr Opin Cell Biol 1: 30–35
Miller DD, Scordilis SP, Hepler PK (1995) Identification and localization of three classes of myosins in pollen tubes ofLilium longiflorum andNicotiana alata. J Cell Sci 108: 2549–2653
Mól R, Matthys-Rochon E, Dumas C (1994) The kinetics of cytological events during double fertilization inZea mays L. Plant J 5: 197–206
Palevitz BA, Tiezzi A (1992) Organization, composition, and function of the generative cell and sperm cytoskeleton. Int Rev Cytol 140: 149–185
Pierson ES, Cresti M (1992) Cytoskeleton and cytoplasmic organization of pollen and pollen tubes. Int Rev Cytol 140: 73–125
Russell SD (1982) Fertilization inPlumbago zeylanica: entry and discharge of the pollen tube into the embryo sac. Can J Bot 60: 2219–2230
— (1992) Double fertilization. Int Rev Cytol 140: 357–388
— (1996) Attraction and transport of male gametes for fertilization. Sex Plant Reprod 9: 337–342
Shimizu T (1995) Role of the cytoskeleton in the generation of spatial patterns inTubifex eggs. Curr Top Devel Biol 31: 197–235
Sonobe S, Shibaoka H (1989) Cortical fine actin filaments in higher plant cells visualized by rhodamine-phalloidin after pretreatment withm-maleimidobenzoic acid N-hydroxysuccinimide ester. Protoplasma 148: 80–86
Swope RE, Kropf D (1993) Pronuclear position and migration during fertilization inPelvetia. Dev Biol 157: 269–276
Valster AH, Hepler PK (1997) Caffeine inhibition of cytokinesis: effect on the phragmoplast cytoskeleton in livingTradescantia stamen hair cells. Protoplasma 196: 155–166
Vos JW, Hepler PK (1998) Calmodulin is uniformly distributed during cell division in living stamen hair cells ofTradescantia virginiana. Protoplasma 201: 158–171
Webb M, Gunning BES (1994) Cell biology of embryo sac development inArabidopsis. In: Williams EG, Clarke AE, Knox RB (eds) Genetic control of self-incompatibility and reproductive development in flowering plants. Kluwer, Dordrecht, pp 461–485
Ye XL, Zee SY, Yeung EC (1997) Suspensor development in the Nun orchid,Phaius tankervilliae. Int J Plant Sci 158: 704–712
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Huang, B.Q., Fu, Y., Zee, S.Y. et al. Three-dimensional organization and dynamic changes of the actin cytoskeleton in embryo sacs ofZea mays andTorenia fournieri . Protoplasma 209, 105–119 (1999). https://doi.org/10.1007/BF01415706
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01415706