Abstract
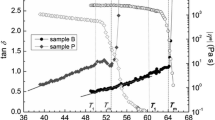
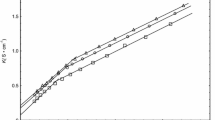
The isothermal ternary phase diagrams for the systems magnesium dodecylsulphate-decanol-water at 40 °C and calcium dodecylsulphate-decanol-water at 50 °C are determined by water deuteron NMR and polarizing microscopic studies. In the magnesium system, three liquid crystalline phases (lamellar and normal and reverse hexagonal) and two isotropic (normal and reverse) solution phases are characterized and their ranges of existence are obtained. The calcium system yields the same liquid crystalline phases, but only the lamellar liquid crystalline phase is investigated in detail. The important observations made are: (i) The lamellar liquid crystalline phase for the magnesium and calcium systems can incorporate, respectively, a maximum of 22.5 and 14.3 mole water per mole surfactant ion against 139 mole water for the corresponding sodium system. (ii) The reverse hexagonal liquid crystalline phase is formed for both the magnesium and calcium systems while no such liquid crystalline phase exists for the corresponding sodium system. (iii) The2H NMR quadrupole splittings obtained in the liquid crystalline phases for C8SO −4 and C12SO −4 surfactant systems with different counterions (Ca2+,Mg2+,Be2+,Na+) reveal that surfactant hydration is almost independent of alkyl chain length and counterions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Khan A, Fontell K, Lindblom G, Lindman B (1982) J Phys Chem 86:4266
Khan A, Fontell K, Lindman B (1984) Mittal KL, Lindman B (eds) Surfactants in Solution, Vol 1, Plenum Press, New York, p 193
Khan A, Fontell K, Lindman B (1984) J Coll Interf Sci 101:193
Khan A, Fontell K, Lindman B (1984) Coll & Surf 11:401
Khan A, Fontell K, Lindman B (1985) Prog Coll & Polym Sci 70:30
Khan A et al. (ed) Magid L, Magnetic Resonance and Scattering in Surfactant Systems, Plenum Press, New York, in press
Khan A, Jönsson B, Wennerström H (1985) J Phys Chem 89:5180
Ekwall P (1975) Brown GH (ed) Advances in Liquid Crystals, Academic Press, New York, London, Vol 1, Chap 1
Tiddy GJT (1980) Phys Rev 57:1
Friberg SE, Osborne DW (1984) Coll & Surf 12:357
Hato M, Shinoda K (1973) J Phys Chem 77:378
Gunnarsson G, Jönsson B, Wennerström H (1980) J Phys Chem 84:3114
Jönsson B, Gunnarsson G, Wennerström H (1982) Mittal KL, Fendler EJ (eds) Solution Behaviour of Surfactants, Plenum Press, New York, p 317
Bratko D, Lindman B (1985) J Phys Chem 89:1437
Wennerström H, Jönsson B, Linse P (1982) J Chem Phys 76:4665
Miyamoto S (1960) Bull Chem Soc Jpn 33:371
Shinoda K, Hirai T (1977) J Phys Chem 81:1842
Rosevear FB (1954) J Am Oil Chem Soc 31:628
Rosevear FB (1968) J soc Cosmet Chem 19:581
Leigh ID, McDonald MP, Wood RM, Tiddy GJT, Trevethan MA (1981) J Chem Soc Farady Trans J 77:2867
Persson NO, Lindman B (1975) J Phys Chem 79:1410
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maciejewska, D., Khan, A. & Lindman, B. Magnesium and calcium surfactants Ternary phase diagrams of magnesium and calcium dodecylsulphate with decanol and water. Colloid & Polymer Sci 264, 909–916 (1986). https://doi.org/10.1007/BF01410643
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01410643