Summary
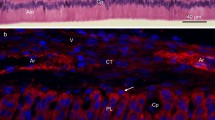
The possibility of using the antisera raised in rabbits against the porcine 25 kDa amelogenin, 32 and 89 kDa enamelins, and the 13–17 kDa nonamelogenin for the differentiation and identification of the protein components in bovine immature enamel was examined. Although the immunoreactivities of these antisera against bovine enamel proteins were weaker than those against the porcine proteins, it was found that these antisera could differentiate and demonstrate immunohistochemically a characteristic distribution of three different kinds of enamel protein components in the bovine secretory stage enamel similar to those observed in the porcine immature enamel. Of the several high molecular weight proteins being reactive to the anti-porcine 32 and 89 kDa enamelin sera, the 130 kDa protein, having the highest molecular weight, was extracted and purified from the bovine enamel sample which was obtained by peeling approximately 30-μm thickness of the outermost layer of the secretory stage enamel. The amino acid composition of the 130 kDa protein was similar to the known bovine enamelins, and was rich in aspartic acid, glutamic acid, proline, and glycine. The results could suggest that the enamelins of lower molecular weight than this protein, which are found in the bovine secretory stage enamel, are derived from this precursor protein.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Snead ML, Lau EC, Zeichner-David M, Fincham AG, Woo SLC, Slavkin HC (1985) DNA sequence for cDNA for murine amelogenin reveals the amino acid sequence for enamel specific protein. Biochem Biophys Res Common 129:812–818
Shimokawa H, Ogata Y, Sasaki S, Sobel ME, McQuillan CI, Tern-line JD, Young MF (1987) Molecular cloning of bovine amelogenin cDNA. Adv Dent Res 1:293–297
Shimokawa H, Tamura M, Ibaraki K, Ogata Y, Sasaki S (1989) Human amelogenin gene. In: Fearnhead RW (ed) Tooth enamel V. Florence Publishers, Yokohama, p 301
Yamakoshi Y, Tanabe T, Fukae M, Shimizu M (1989) Amino acid sequence of 25 kDa amelogenin. In: Fearnhead RW (ed) Tooth enamel V. Florence Publishers, Yokohama, p 314
Termine JD, Belcourt AB, Christner PJ, Conn KM, Nylen MU (1980) Properties of dissociatively extracted fetal tooth matrix proteins. 1. Principal molecular species in developing bovine enamel. J Biol Chem 255:9760–9768
Deutsch D, Palmon A, Fisher LW, Kolodny N, Termine JD, Young MF (1991) Sequencing of bovine enamelin (“Tuftelin”): a novel acidic enamel protein. J Biol Chem 266:16021–16028
Fukae M, Tanabe T (1987) Nonamelogenin components of porcine enamel in the protein fraction free from the enamel crystals. Calcif Tissue Int 40:286–293
Uchida T, Tanabe T, Fukae M, Shimizu M (1991) Immunocytochemical and immunochemical detection of a 32 kDa nonamelogenin and related proteins in porcine tooth germs. Arch Histol Cytol 54:527–538
Rosenbloom J, Lally E, Dixon M, Spencer A, Herold R (1986) Production of a monoclonal antibody to enamelins which does not cross-react with amelogenins. Calcif Tissue Int 39:412–415
Fukae M, Shimizu M (1974) Studies on the proteins of developing bovine enamel. Arch Oral Biol 19:381–386
Uchida T, Tanabe T, Fukae M, Shimizu M, Yamada M, Miyake K, Kobayashi S (1991) Immunochemical and immunohistochemical studies, using antisera against porcine 25 kDa amelogenin, 89 kDa enamelin and the 13–17 kDa nonamelogenins, on immature enamel of the pig and rat. Histochemistry 96:129–138
Tanabe T, Fukae M, Uchida T, Shimizu M (1992) The localization and characterization of proteinases for the initial cleavage of porcine amelogenin. Calcif Tissue Int 51:213–217
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Towbin H, Staehelin T, Gordon J (1974) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Hsu SM, Aine LR, Fanger H (1981) A comparative study of the perioxidase-antiperioxidase method and an avidin-biotin complex method for studying polypeptide hormones with radioimmunoassay antibodies. Am J Clin Pathol 75:734–738
Ogata Y, Shimokawa H, Sasaki S (1988) Purification, characterization and biosynthesis of bovine enamelins. Calcif Tissue Int 43:389–397
Takagi T, Suzuki M, Baba T, Minegishi K, Sasaki S (1984) Complete amino acid sequence of amelogenin in developing bovine enamel. Biochem Biophys Res Commun 121:592–597
Shimokawa H, Sobel ME, Sasaki M, Termine JD, Young MF (1987) Heterogeneity of amelogenin mRNA in the bovine tooth germ. J Biol Chem 262:4042–4047
Aoba T, Shimoda S, Shimokawa H, Inage T (1992) Common epitopes of mammalian amelogenins at the C-terminus and possible functional roles of the corresponding domain in enamel mineralization. Calcif Tissue Int 51:85–91
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fukae, M., Tanabe, T., Uchida, T. et al. Enamelins in the newly formed bovine enamel. Calcif Tissue Int 53, 257–261 (1993). https://doi.org/10.1007/BF01320911
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01320911