Summary
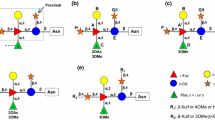
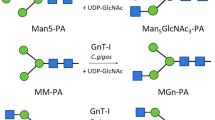
The α-glucosidase inhibitor bromoconduritol inhibits the formation of the N-linked, complex-type oligosaccharides of the glycoproteins from influenza viruses (fowl plague virus, influenza virus PR-8) and from sindbis virus. Viral glycoproteins produced in bromoconduritol-treated chickenembryo and baby-hamster kidney cells are fully glycosylated, but accumulate N-linked, high-mannose oligosaccharides of the composition Glc1Manx (GlcNAc)2 (x=7, 8, and 9). Other α-glucosidase inhibitors (nojirimycin, deoxynojirimycin, acarbose) were not specific inhibitors of oligosaccharide processing under the conditions used in the present investigation.
In bromoconduritol-treated, sindbis virus-infected chicken-embryo and baby-hamster kidney cells, the sindbis glycoproteins are metabolically stable. Specific proteolytic cleavage of the polyprotein precursors to form E2 and E1 occurs in bromoconduritol-treated chicken-embryo cells, but cleavage of PE2 to E2 is prevented in the infected baby-hamster kidney cells. Yet, release of infectious sindbis virus particles is inhibited in both cell types indicating that the formation of complex oligosaccharides is required for a late step in virus formation.
The release of virus particles from influenza virus PR-8-infected bromoconduritol-treated chicken-embryo cells is not inhibited, and virus with only high-mannose oligosaccharides is formed. In contrast, when chickenembryo cells were infected with the influenza virus fowl plague virus, release of infectious particles was inhibited. The fowl plague virus hemagglutinin is cleaved in chicken-embryo cells, in contrast to the hemagglutinin of the PR-8 virus. However, the cleavage products HA1 and HA2 do not reach the cell surface. In addition, or as a consequence, HA1 and HA2 are proteolytically broken down, whereas uncleaved hemagglutinin of PR-8 appeared metabolically stable. These results may explain the decrease in formation of fowl plague virus particles and the lack of effect on PR-8 virus in bromoconduritol-treated cells. This work thus shows different biological roles for oligosaccharide processing.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beyer, T. A., Sadler, J. E., Rearick, J. I., Paulson, J. C., Hill, R. L.: Glycosyltransferases and their use in assessing oligosaccharide structure and structure-function relationships. Adv. Enzymol.52, 23–175 (1981).
Bosch, F. X., Garten, W., Klenk, H.-D., Rott, R.: Proteolytic cleavage of influenza virus hemagglutinins: Primary structure of the connecting peptide between HA1 and HA2 determines proteolytic cleavability and pathogenicity of avian influenza viruses. Virology113, 725–735 (1981).
Bosch, J. V., Schwarz, R. T., Ziemiecky, A., Friis, R. R.: Oligosaccharide modifications and the site of processing of gPr 92env, the precursor for the viral glycoprotein of rous sarcoma virus. Virology119, 122–132 (1982).
Bracha, M., Schlesinger, M. J.: Defects in RNA+ temperature-sensitive mutants of sindbis virus and evidence for a complex of PE2 E1 viral glycoproteins. Virology74, 441–445 (1976).
Burke, D. J., Keegstra, K.: Purification and composition of the proteins from sindbis virus grown in chick and BHK-cells. J. Virol.20, 676–686 (1976).
Caliguiri, L. A., Tamm, I.: The role of cytoplasmic membranes in polio virus biosynthesis. Virology42, 100–111 (1970).
Datema, R., Schwarz, R. T., Jankowski, A. W.: Fluoroglucose-inhibition of protein glycosylationin vivo. Inhibition of mannose and glucose incorporation into lipid-linked oligosaccharides. Eur. J. Biochem.109, 331–341 (1980a).
Datema, R., Schwarz, R. T., Winkler, J.: Glycosylation of influenza virus proteins in the presence of fluoroglucose occurs via a different pathway. Eur. J. Biochem.110, 355–366 (1980b).
Datema, R., Schwarz, R. T.: Effect of energy depletion on the glycosylation of a viral glycoprotein. J. Biol. Chem.256, 11191–11198 (1981).
Datema, R., Romero, P. A., Legler, G., Schwarz, R. T.: Inhibition of formation of complex oligosaccharides by the glucosidase inhibitor bromoconduritol. Proc. Natl. Acad. Sci. U.S.A.79, 6787–6791 (1982).
Elbein, A. D., Dorling, P. R., Vosbeck, K., Horisberger, M.: Swainsonine prevents the processing of the oligosaccharide chains of influenza virus hemagglutinin. J. Biol. Chem.257, 1573–1576 (1982).
Grinna, L. S., Robbins, P. W.: Glycoprotein biosynthesis. Rat liver microsomal glucosidases which process oligosaccharides. J. Biol. Chem.254, 8814–8818 (1979).
Hetkamp, H., Bause, E., Legler, G.: Calf liver microsomal glucosidases: characterization and inhibition by various inhibitors. Hoppe-Seyler's Zeitschr. Physiol. Chem.363, 1019 (1982).
Hakimi, J., Atkinson, P. H.: Glycosylation of intracellular sindbis virus glycoproteins. Biochemistry21, 2140–2145 (1982).
Klenk, H.-D., Rott, R.: Cotranslational and posttranslational processing of virus glycoproteins. Curr. Top. Microbiol. Immunol.90, 19–48 (1980).
Klenk, H.-D., Rott, R., Orlich, M., Blödorn, J.: Activation of influenza A viruses by trypsin treatment. Virology68, 426–439 (1975).
Kornfeld, S.: Oligosaccharide processing during glycoprotein biosynthesis. In:Horowitz, M. I. (ed.), The glycoconjugates, Vol. III. New York: 1982.
Kornfeld, S., Gregory, W., Chapman, A.: Class-E-Thy-1-negative mouse lymphona cells utilize an alternate pathway of oligosaccharide processing to synthesize complex-type oligosaccharides. J. Biol. Chem.259, 11649–11654 (1979).
Legler, G., Lotz, W.: Untersuchungen zum Wirkungsmechanismus glycosidspaltender Enzyme VII. Funktionelle Gruppen am aktiven Zentrum einer α-Glucosidase aus Sacch. cerevisiae. Hoppe-Seyler's Zeitschr. Physiol. Chem.354, 243–254 (1973).
Legler, G.: Glucosidases. Meth. Enzymol.46, 368–381 (1977).
Li, E., Tabas, I., Kornfeld, S.: The synthesis of complex-type oligosaccharides. I. Structure of the lipid-linked oligosaccharide precursor of the complex-type oligosaccharides of the vesicular stomatitis virus G protein. J. Biol. Chem.253, 7762–7770 (1978).
Lohmeyer, J., Talens, L. T., Klenk, H.-D.: Biosynthesis of the influenza virus envelope in abortive infection. J. gen. Virol.42, 73–88 (1979).
Robbins, P. W., Hubbard, S. C., Turco, S. J., Wirth, D. F.: Proposal for a common oligosaccharide intermediate in the synthesis of membrane glycoproteins. Cell12, 893–900 (1977).
Romero, P. A., Datema, R., Schwarz, R. T.: N-Methyl-1-deoxynojirimycin, a novel inhibitor of glycoprotein processing, and its effects on fowl plague virus maturation. Virology130, 238–242 (1983).
Saunier, B., Kilker, R. D., Tkacz, J. S., Quaroni, A., Herscovics, A.: Inhibition of N-linked complex oligosaccharide formation by 1-deoxy-nojirimycin, an inhibitor of processing glucosidases. J. Biol. Chem.257, 14155–14161 (1982).
Schlesinger, M. J., Malfer, C.: Cerulenin blocks fatty acid acylation of glycoproteins and inhibits vesicular stomatitis and sindbis virus particle formation. J. Biol. Chem.257, 9887–9890 (1982).
Schwarz, R. T., Rohrschneider, J. M., Schmidt, M. F. G.: Suppression of glycoprotein formation of semliki forest, influenza, and avian sarcoma viruses by tunicamycin. J. Virol.19, 782–791 (1976).
Schwarz, R. T., Datema, R.: The lipid pathway of protein glycosylation and its inhibitors. The biological significance of protein-bound carbohydrates. Adv. Carbohyd. Chem. Biochem.40, 287–379 (1982a).
Schwarz, R. T., Datema, R.: Inhibition of the dolichol pathway of protein glycosylation. Meth. Enzymol.83, 432–443 (1982b).
Sefton, B. M.: Immediate glycosylation of sindbis virus membrane proteins. Cell10, 659–668 (1977).
Simons, K., Garoff, H.: The budding mechanisms of enveloped animal viruses. J. gen. Virol.50, 1–21 (1980).
Tartakoff, A. M.: The Golgi complex: crossroads for vesicular traffic. Int. Rev. Exp. Pathol.22, 227–251 (1980).
Truscheit, E., Frommer, W., Junge, B., Müller, L., Schmidt, D. D., Wingender, W.: Chemie und Biochemie mikrobieller α-Glucosidase-Inhibitoren. Angewandte Chemie93, 738–755 (1981).
Tulsiani, D. R. P., Hubbard, S. C., Robbins, P. W., Touster, O.: α-Mannosidases of rat liver Golgi membranes. J. Biol. Chem.257, 3660–3668 (1982).
Turco, S. J., Pickard, J. L.: Altered G-protein glycosylation in vesicular stomatitis virus-infected, glucose deprived baby hamster kidney cells. J. Biol. Chem.257, 8674–8679 (1982).
Welch, W. J., Sefton, B. M.: Two small virus specific polypeptides are produced during infection with sindbis virus. J. Virol.29, 1186–1195 (1979).
Zimmerman, T., Schäfer, W.: Effect of p-fluorophenylalanine on fowl plague virus multiplication. Virology11, 676–698 (1960).
Author information
Authors and Affiliations
Additional information
With 6 Figures
Rights and permissions
About this article
Cite this article
Datema, R., Romero, P.A., Rott, R. et al. On the role of oligosaccharide trimming in the maturation of sindbis and influenza virus. Archives of Virology 81, 25–39 (1984). https://doi.org/10.1007/BF01309294
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01309294