Summary
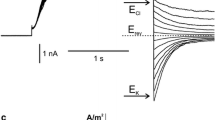
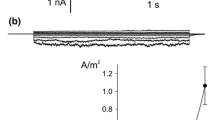
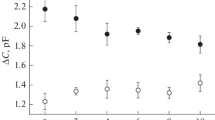
The cytoplasmic drop formed of effused cytoplasm fromChara internodes is enclosed by a membrane. Patch clamp experiments have been carried out on this membrane, revealing a K+ channel as the most frequently detected ion translocator. The K+ channel is saturated at a level of about 20 pA inward and 10 pA outward current. The channel conductance is dependent on the accessability of K+ ions, its maximum value amounts to about 165 pS. The discrimination of Na+ and Cl− is significant, permeability ratios PNa/PK and PCl/PK were estimated to be 0.01 either. Binding experiments with the fluorescent probe concanavalin A/FITC suggest that the membrane is derived from the tonoplast.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- EK :
-
K+ equilibrium potential
- FITC:
-
fluorescein isothiocyanat
- Vm :
-
membrane voltage
- Vpip :
-
pipette clamp voltage
- Vr :
-
reversal voltage
References
Bentrup FW, Hoffmann B, Gogarten-Boekels M, Gogarten JP (1985) A patch clamp study of tonoplast electrical properties in vacuoles isolated fromChenopodium rubrum suspension cells. Z Naturforsch 40c: 886–890
Hamill OP, Marty A, Neher E, Sakmann B, Sieworth FJ (1981) Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflügers Arch 391: 85–100
Inoue I, Ishida N, Kobatake Y (1973) Studies of excitable membrane formed on the surface of protoplasmic drops isolated fromNitella. IV. Excitability of the drop membrane in various compositions of the external salt solution. Biochim Biophys Acta 330: 27–38
Kamiya N, Kuroda K (1957 a) Cell operation inNitella. I. Cell amputation and effusion of the endoplasm. Proc Jpn Acad 33: 149–152
— — (1957 b) Cell operation inNitella. II. Behaviour of isolated endoplasm. Proc Jpn Acad 33: 201–205
Kolb H-A (1984) Measuring the properties of single channels in cell membranes. In:Stein WD (ed) Current topics in membranes and transport, vol 21. Academic Press, London
Koppenhöfer E, Schramm M (1974) A method for electrical measurements on isolated protoplasmic droplets fromNitella. Pflügers Arch 350: 341–346
Krawczyk S (1978) Ionic channel formation in a living cell membrane. Nature 273: 56–57
Moran N, Ehrenstein G, Iwasa K, Bare C, Mischke C (1984) Ion channels in plasmalemma of wheat protoplasts. Science 226: 835–838
Neher E (1982) Unit conductance studies in biological membranes. In:Baker PF (ed) Techniques in cellular physiology. Elsevier, Amsterdam
Okazaki Y, Shimmen T, Tazawa M (1984) Turgor regulation in a brackish Charophyte,Lamprothamnium succinctum. II. Changes in K+, Na+ and Cl− concentrations, membrane potential and membrane resistance during turgor regulation. Plant Cell Physiol 25: 573–581
Raven JA (1976) Transport in algal cells. In:Lüttge U, Pitman MG (eds) Encyclopedia of plant physiology, NS, vol 2 A. Springer, Berlin Heidelberg New York
Reeves M, Shimmen T, Tazawa M (1985) Ionic activity gradients across the surface membrane of cytoplasmic droplets prepared fromChara australis. Plant Cell Physiol 26: 1185–1193
Sachs F (1983) Is the acetylcholine receptor a unit-conductance channel? In:Sakmann B, Neher E (eds) Single-channel recording. Plenum Press, New York
Sakano K, Tazawa M (1985) Minivacuole formation in the protoplasmic droplets from internodal cells ofChara. Cell Struct Funct 10: 499
— — (1986) Tonoplast origin of the envelope membrane of cytoplasmic droplets prepared fromChara internodal cells. Protoplasma 131: 247–249
Sakmann B, Neher E (1983) Geometric parameters of pipettes and membrane patches. In:Sakmann B, Neher E (eds) Single channel recording. Plenum Press, New York
Schroeder JI, Hedrich R, Fernandez JM (1984) Potassium-selective single channels in guard cell protoplasts ofVicia faba. Nature 312: 361–362
Svintitskikh VA, Andrianov VK, Bulychev AA (1985) Photo-induced H+ transport between chloroplasts and the cytoplasm in a protoplasmic droplet ofCharaceae. J Exp Bot 36: 1414–1429
Tazawa M, Kikuyama M, Shimmen T (1976) Electric characteristics and cytoplasmic streaming ofCharaceae cells lacking tonoplast. Cell Struct Funct 1: 167–176
Ueda T, Muratsugu M, Inoue I, Kobatake Y (1974) Structural changes of excitable membrane formed on the surface of protoplasmic drops isolated fromNitella. J Membr Biol 18: 177–186
Zimmermann U, Steudle E (1978) Generation of action potentials inChara corallina by turgor pressure changes. Planta 138: 173–179
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lühring, H. Recording of single K+ channels in the membrane of cytoplasmic drop ofChara australis . Protoplasma 133, 19–28 (1986). https://doi.org/10.1007/BF01293183
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01293183