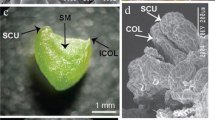
Abstract
Excised seedling leaf segments of winged bean [Psophocarpus tetragonolobus (L.) DC.] underwent direct somatic embryogenesis under appropriate incubation conditions. Initiation and development of the somatic embryos occurred using a two-step culture method. The culture procedure involved incubation for 28 days on MS basal medium supplemented with 0.1–0.5 mg/l NAA and 1.0–2.0 mg/l BA (induction medium) before transfer to MS medium supplemented with 0.1 mg/l IAA and 2.0 mg/l BA (embryo development medium). The initial exposure to low levels of NAA coincident with high levels of BA in the induction medium was essential for embryogenic induction. Maximum embryogenesis (43.3%) was obtained with 0.2 mg/l NAA and 2.0 mg/l BA, and at least 14 days on induction medium were required prior to transfer to the embryo development medium. The conversion frequency of cotyledonary embryos was 53.3% upon culture on MS medium containing 0.1 mg/l ABA for 7 days followed by transfer to MS medium supplemented with 0.1 mg/l IBA and 0.2 mg/l BA. Following conversion, the regenerated plantlets were transferred to soil and showed normal morphological characteristics.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- MS :
-
Murashige and Skoog (1962) medium
- 2,4-D :
-
2,4-dichlorophenoxyacetic acid
- NAA :
-
1-naphthaleneacetic acid
- IAA :
-
indole-3-acetic acid
- IBA :
-
indole-3-butyric acid
- BA :
-
6-benzylaminopurine
- ABA :
-
abscisic acid
References
Ahmed R, Dutta Gupta S, De DN (1996) Somatic embryogenesis and plant regeneration from leaf derived callus of winged bean [Psophocarpus tetragonolobus (L.) Dc.] Plant Cell Rep 15:531–535
Baker CM, Wetzstein HY (1992) Somatic embryogenesis and plant regeneration from leaflets of PeanutArachis hypogaea. Plant Cell Rep 11:71–75
Baker CM, Wetzstein HY (1994) Influence of auxin type and concentrations on peanut somatic embryogenesis. Plant Cell Tiss Org Cult 36:361–368
Barna KS, Wakhlu AK (1993) Somatic embryogenesis and plant regeneration from callus cultures of chickpea (Cicer arietinum L.). Plant Cell Rep 12:521–524
Barna KS, Wakhlu AK (1995) Direct somatic embryogenesis and plantlet regeneration from immature leaflets in chickpea. In Vitro Cell Dev Biol 31P:137–139
Buchheim JA, Colburn SM, Ranch SP (1989) Maturation of soybean somatic embryos and the transition to plantlet growth. Plant Physiol 89:768–775
Chengalrayan K, Sathaye SS, Hazra S (1994) Somatic embryogenesis from mature embryo-derived leaflets of peanut (Arachis hypogea L.). Plant Cell Rep 13:578
Christianson ML, Warnick DA (1985) Temporal requirement for phytohormone balance in the control of organogenesis. In Vitro Dev Biol 112:494–497
Finstad K, Brown DCB, Joy K (1993) Characterization of competence during induction of somatic embryogenesis in alfalfa tissue culture. Plant Cell Tis Org Cult 34:125–132
Gregory HM, Haq N, Evans PK (1980) Regeneration of plantlets from leaf callus of the winged beanPsophocarpus tetragonolobus (L) DC. Plant Sci Lett 18:395–400
Hazara S, Sathaye SS, Mascarenhas AF (1989) Somatic embryogenesis in peanut (Arachis hypogaea). Biotechnology 7:949–951
Lazzeri PA, Hildebrand DF, Collins GB (1987) Soybean somatic embryogenesis. Effects of hormones and culture manipulations. Plant Cell Tiss Org Cult 10:197–208
Maheswaren G, Williams EG (1984) Direct somatic embryoid formation on immature embryos ofTrifolium repens, T, pratense, andMedicago sativa, and rapid clonal propagation ofT. repens. Ann Bot 54:201–211
Malik KA, Saxena PK (1992) Somatic embryogenesis and shoot regeneration from intact seedlings ofPhaseolus acutifolius. P. aures (L) Wilczek,P. coccineus L., andP. wrightii L. Plant Cell Rep 11:163–168
McKently A (1991) Direct somatic embryogenesis from axes of mature peanut embryos. In Vitro Cell Dev Biol 27P:197–200
Mehta U, Mohan Ram HY (1981) Tissue culture and whole plant regeneration in the winged bean (Psophocarpus tetragonolobus L.). Ann Bot 47:163–166
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
NAS Report (1979) The winged bean. A high protein crop for the tropics. National Academy of Sciences, Washington, D.C.
Ozias-Akins P (1989) Plant regeneration from immature embryos of peanut. Plant Cell Rep 8:217–218
Parrott WA, Bailey MA, Durham RE, Mathews HV (1992) Tissue culture and regeneration in legumes. In: Moss JP (eds) Biotechnology and crop improvement in Asia. ICRISAT, Patancheru, India, pp 115–148
Sellers RM, Southward GM, Phillips GC (1990) Adventitious somatic embryogenesis, from cultured immature zygotic embryos of peanut using soybean as a model system. Crop Sci 30:408–414
Suhasini K, Sagare AP, Krishnamurthy KV (1994) Direct somatic embryogenesis from mature embryo axes in chickpea (Cicer arietinum L.) Plant Sci 102:189–194
Tran Thanh Van K, Lie-Schricke H. Marcotte JL, Trinh TH (1986) Winged bean [Psophocarpus tetragonolobus (L.) DC.]. In: Bajaj YPS (eds) Biotechnology in agriculture and forestry, vol2: crop 1. Springer, Berlin Heidelberg New York, pp 556–567
Venketeswaran S, Dias MADL, Weyers UV (1992) Organogenesis and somatic embryogenesis from callus of winged bean [Psophocarpus tetragonolobus (L.) DC.] Acta Hortic 280:202–206
Wetzstein HY, Baker CM (1993) The influence of somatic embryo morphology and histology on germination and conversion in peanut. Plant Sci 92:81–89
Wilson VM, Haq N, Evans PK (1985) Protoplast isolation culture and plant regeneration in the winged bean.Psophocarpus tetragonolobus (L.) DC. Plant Sci 41:61–68
Author information
Authors and Affiliations
Additional information
Communicated by G. Phillips
Rights and permissions
About this article
Cite this article
Dutta Gupta, S., Ahmed, R. & De, D.N. Direct somatic embryogenesis and plantlet regeneration from seedling leaves of winged bean,Psophocarpus tetragonolobus (L.) DC. Plant Cell Reports 16, 628–631 (1997). https://doi.org/10.1007/BF01275504
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01275504