Summary
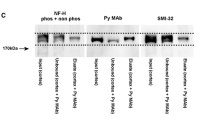
During antibody screening on sections of rat cerebellum, we noticed a group of small neurons which exhibited unusual staining properties. They were robustly immunopositive for the high molecular weight neurofilament protein, moderately immunostained with antibodies to the low molecular weight neurofilament protein and α-internexin, but only faintly immunoreactive (in PAP sections) or essentially immunonegative (in immunofluorescent sections) with all members of a panel of antibodies directed against the middle molecular weight neurofilament protein. Since neurons generally react equally well with phosphate-independent, (antibodies to) low, middle and high molecular weight neurofilament protein, we conclude that middle molecular weight neurofilament protein is present in these cells in an unusually low relative amount. These cells are found in the granular layer and appear concentrated in the flocculus, ventral paraflocculus, and vermis, particularly in the ventral uvula and nodulus (lobules IXd and X). Previous studies performed by Hockfield defined a population of neurons of similar appearance and distribution using the monoclonal antibody Rat-302, which recognized an uncharacterized 160 kDa protein. We show here that the cells described by Hockfield are identical to those we have found and furthermore that the Rat-302 antibody specifically recognizes the dephosphorylated form of the lysine-serine-proline repeated sequences of high molecular weight neurofilament protein. These cells were studied by pre-embedding immunoelectron microscopy. The nucleus is deeply indented and shows little condensed chromatin. The cytoplasm contains scattered microtubules and a larger number of neurofilaments than expected in a small cell. There are numerous large dense core vesicles, an unusual organelle consisting of ringlet subunits, and relatively little granular endoplasmic reticulum. A thin axon and a single stout dendritic trunk emanate from the perikaryon. Although the cell body and the dendritic shaft may form either complex contacts with mossy fibres (resembling those previously termed en marron synapses) or simple symmetric synapses with small boutons containing pleomorphic vesicles, most of the synaptic relations are established on the shafts of brush-like branchlets that form at the tip of the dendrite and enter one or two glomeruli. Each branchlet forms an extraordinarily extensive asymmetric synapse with the mossy fibre rosette and the subsynaptic region shows a microfibrillar web connected to the postsynaptic density. In addition to other organelles, the branchlets contain numerous mitochondria and large dense core vesicles. Short, non-synaptic appendages with few cytoplasmic organelles emanated from the cell body, dendritic shaft and branchlets. The immunoreaction products of all neurofilament antibodies were similarly distributed within the small cells, and were absent from the granular reticulum, the Golgi apparatus, the appendages and the subsynaptic region. These high molecular weight neurofilament protein rich small cells correspond to the pale cells, the calretinin and secretogranin positive small cells and the unipolar brush neurons newly described with the Golgi method. Unlike the multipolar Golgi neurons, unipolar brush cells are not immunopositive for the inhibitory neurotransmitters GABA or glycine. The unusual concentration of secretogratin and two different types of calcium-binding protein (calretinin and high molecular weight neurofilament protein) along with the high content of mitochondria suggest that these cells subserve a function that requires an unusual degree of metabolic activity, perhaps as a result of their unusually rich synaptic connections.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altman, J. &Bayer, S. A. (1977) Time of origin and distribution of a new cell type in the rat cerebellar cortex.Experimental Brain Research 29, 265–74.
Barmack, N. H., Baughman, R. W. &Eckenstein, F. P. (1992a) Cholinergic innervation of the cerebellum of rat, rabbit, cat and monkey revealed by choline acetyl-transferase activity and immunohistochemistry.Journal of Comparative Neurology 317, 233–49.
Barmack, N. H., Baughman, R. W., Eckenstein, F. P. &Shojaku, H. (1992b) A secondary vestibular cholinergic projection to the cerebellum of rabbit and rat as revealed by choline acetyltransferase immunohistochemistry, retrograde and orthograde tracers.Journal of Comparative Neurology 317, 250–70.
Brodal, A. &Drabløs, P. A. (1963) Two types of mossy fiber terminals in the cerebellum and their regional distribution.Journal of Comparative Neurology 121, 173–88.
Cajal, S. Ramón Y (1911)Histologie du Systeme nerveux de l'Homme et des Vertébrés (translated byL. Azoulay) Volumes I and II. Paris: Maloine.
Carden, M. J., Trojanowski, J. Q., Schlaepfer, W. W. &Lee, V. M.-Y. (1987) Two-stage expression of neurofilament polypeptides during rat neurogenesis with early establishment of adult phosphorylation patterns.Journal of Neuroscience 7, 3489–504.
Chiu, F. C., Barnes, E. A., Das, K., Haley, J., Socolow, P., Macaluso, F. P. &Fant, J. (1989) Characterization of a novel 66 kd subunit of mammalian neurofilaments.Neuron 2, 1435–45.
Cozzi, M. G., Rosa, P., Greco, A., Hille, A., Huttner, W. B., Zanini, A. &De Camieli, P. (1989) Immunohistochemical localization of secretogranin II in the rat cerebellum.Neuroscience 28, 423–4.
Draeger, U. C., Edwards, D. L. &Keeinschmidt, J. (1983) Neurofilaments contain α-melanocyte-stimulating hormone (α-MSH)-like immunoreactivity.Proceedings of the National Academy of Sciences (USA) 80, 6408–12.
Eaker, E., Shaw, G. &Sninsky, C. (1990) Neurofilament immunoreactivity in myenteric neurons differs from that found in the central nervous system.Gastroenterology 99, 1364–71.
Eccles, J. C., Ito, M. &Szentágothai, J. (1967)The Cerebellum as a Neuronal Machine. New York: Springer Verlag.
Fliegner, K. H. &Liem, R. K. H. (1991) Cellular and molecular biology of intermediate filaments.International Reviews of Cytology 131, 109–67.
Fliegner, K. H., Ching, G. Y. &Liem, R. K. H. (1990) The predicted amino acid sequence of α-internexin is that of a novel neuronal intermediate filament protein.EMBO Journal 9, 749–55.
Floris, A., Dino, M., Jacobowitz, D. M. &Mugnaini, E. (1994) The unipolar brush cells of the mammalian cerebellar cortex studied by calretinin immunocytochemistry: light and electron microscopy.Journal of Neurosdence, in press.
Floris, A., Dunn, M. E., Berrebi, A. S., Jacobowitz, D. M. &Mugnaini, E. (1992) Pale cells of the flocculonodular lobe are calretinin positive.Society for Neurosdence Abstracts 22, 853.
Fox, C. A., Hillman, D. E., Siegesmund, K. A. &Dutta, C. R. (1967) The primate cerebellar cortex: a Golgi and electron microscopic study.Progress in Brain Research 25, 174–225.
Gabbott, P. L. A., Somogyi, J., Stewart, M. G. &Hámori, J. (1986) GABA-immunoreactive neurons in the rat cerebellum: a light and electron microscope study.Journal of Comparative Neurology 251, 474–90.
Geneser-Jensen, F. A. &Blackstad, T. W. (1973) Distribution of acetylcholinesterase in the hippocampal region of the guinea pig.Zeitschrift für Zettforschung 114, 460–81.
Golgi, C. (1882) Sulla fina anatomia degli organi centrali del sistem a nervoso. I. Note preliminari sulla struttura, morfologia e vicendevoli rapporti delle cellule gangliari.Archives Italiennes de Biologie 3, 285–99.
Golgi, C. (1883) Sulla fina anatomia degli organi centrali del sistema nervoso. IV. Sulla fina anatomia delle circonvoluzion cerebellari.Archives Italiennes de Biologie 4, 92–123.
Hámori, J. &Szentágothai, J. (1966) Participation of Golgi neuron processes in the cerebellar glomeruli: an electron microscope study.Experimental Brain Research 186, 454–7.
Harris, J., Ayyub, C. &Shaw, G. (1991) A molecular dissection of the carboxyterminal tails of the major neurofilament subunits NF-M and NF-H.Journal of Neuroscience Research 30, 47–62.
Hockfield, S. (1987) A Mab to a unique cerebellar neuron generated by immunosuppression and rapid immunization.Science 237, 67–70.
Ito, M. (1984)The Cerebellum and Neural Control. New York: Raven Press.
Jakob, A. P. (1928) Das Kleinhirn. InHandbuch der mikroskopischen Anatomie des Menschen. (edited byVon Möllendorff, W.) Vol. IV/1 pp. 674–916. Berlin: Springer Verlag.
Kaplan, M. P., Chin, S. S. M., Fliegner, K. H. &Liem, R. K. H. (1990) α-Internexin, a novel neuronal intermediate filament protein precedes the low molecular weight neurofilament protein (NF-L) in the developing rat brain.Journal of Neuroscience 10, 2735–48.
Lange, W. (1974) Regional differences in the distribution of Golgi cells in the cerebellar cortex of man and some other mammals.Cell and Tissue Research 153, 219–26.
Mugnaini, E. (1972) The histology and cytology of the cerebellar cortex. InThe Comparative Anatomy and Histology of the Cerebellum. The Human Cerebellum, Cerebellar Connections, and Cerebellar Cortex. (edited byLarsell, O. &Jansen, J.) pp. 201–64. Minneapolis: The University of Minnesota Press.
Mugnaini, E. &Floris, A. (1993) The unipolar brush cell: a neglected neuron of the mammalian cerebellar cortex.Journal of Comparative Neurology. In press.
Mugnaini, E. &Floris, A. (1994) The extraordinary synapses of the cerebellar unipolar brush cell of the rat cerebellum.Synapse. In press.
Osen, K. K. &Roth, K. (1969) Histochemical localization of cholinesterases in the cochlear nuclei of the cat, with notes on the origin of acetylcholinesterase-positive afferents and the superior olive.Brain Research 16, 165–85.
Ottersen, O. P., Storm-Mathisen, J. &Somogyi, P. (1988) Colocalization of glycine-like and GABA-like immunoreactivities in Golgi cell terminals in the rat cerebellum: a postembedding light and electron microscopic study.Brain Research 450, 342–53.
Palay, S. L. &Chan-Palay, V. (1974)Cerebellar Cortex, Cytology and Organization. Berlin: Springer-Verlag.
Retzius, G. (1982) Kleinere Mittheilungen von dem Gebiete der Nervenhistologie. I. Ueber die Golgi'schen Zellen und die Kletterfasern Ramón y Cajal's in der Kleinhirnrinde. InBiologische Untersuchungen. Neue Folge, Vol. IV pp. 57–9. Stockholm: Samson and Wallin.
Sahin, M. &Hockfield, S. (1990) Molecular identification of the Lugaro cell in the cat cerebellar cortex.Journal of Comparative Neurology 301, 575–84.
Scott, D., Smith, K. E., O'brien, B. J. &Angelides, K. J. (1985) Characterization of mammalian neurofilament triplet proteins: subunit stoichiometry and morphology of native and reconstituted filaments.Journal of Biological Chemistry 260, 10736–47.
Shaw, G. (1991) Neurofilament proteins. InThe Neuronal Cytoskeleton (edited byBurgoyne, R. D.) pp. 185–214. New York: Wiley-Liss.
Shaw, G. (1992) A neurofilament-specific sequence motif.Trends in Biological Sciences 17, 345.
Shaw, G., Fischer, S. &Weber, K. (1985) α-MSH and neurofilament M-protein share a continuous epitope but not extended sequences.FEBS Letters 181, 343–6.
Shaw, G., Osborn, M. O. &Weber, K. (1986) Reactivity of a panel of neurofilament antibodies on phosphorylated and dephosphorylated neurofilaments.European Journal of Cell Biology 42, 1–9.
Shaw, G., Osborn, M. O. &Weber, K. (1981) An immunofluorescence microscopical study of the neurofilament triplet proteins, vimentin and glial fibrillary acidic protein in the adult rat brain.European Journal of Cell Biology 26, 68–82.
Shaw, G. &Weber, K. (1982) Differential expression of the neurofilament triplet proteins in brain development.Nature 298, 277–9.
Steinert, P. M. &Roop, D. R. (1988) Molecular and cellular biology of intermediate filaments.Annual Review of Biochemistry 67, 593–625.
Sternberger, L. A. &Sternberger, N. H. (1983) Monoclonal antibodies distinguish phosphorylated and non-phosphorylated forms of neurofilamentsin situ.Proceedings of the National Academy of Sciences (USA) 80, 6126–30.
Sturrock, R. R. (1990) A quantitative histological study of Golgi II neurons and pale cells in different cerebellar regions of the adult and ageing mouse brain.Zeitschrift für mikroskopische anatomische Forschung 104, 705–14.
Trojanowski, J. Q., Walkenstein, N. &Lee, V. M.-Y. (1986) Expression of neurofilament subunits in neurons of the central and peripheral nervous system: an immunohistochemical study with monoclonal antibodies.Journal of Neuroscience 6, 650–60.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Harris, J., Moreno, S., Shaw, G. et al. Unusual neurofilament composition in cerebellar unipolar brush neurons. J Neurocytol 22, 1039–1059 (1993). https://doi.org/10.1007/BF01235748
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01235748