Summary
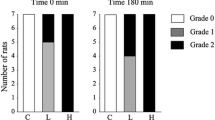
Isolated rat islets were maintainedin vitro in a perifusion system, exposed to alloxan (20 mg/100 ml) for 5 minutes in the presence of agents which affect cAMP metabolism and subsequently stimulated with glucose. The rate of insulin secretion was monitored throughout the period of perifusion. Exposure to alloxan alone produces complete inhibition of glucose-induced insulin release [18] whereas concomitant exposure to caffeine and theophylline for this brief interval provided almost complete protection of the islets from the inhibitory action of alloxan. Glucagon, cAMP and DBcAMP did not protect the islets from alloxan. Pre-treatment of the islets with either theophylline or glucagon and DBcAMP did not provide protection. These findings indicate that the protective action of theophylline and caffeine against alloxan is unrelated to the effect of these agents on cAMP metabolism in the beta cell.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bhattacharya, G.: Protection against alloxan diabetes by mannose and fructose. Sience117, 230–231 (1953)
Blecher, M.: Biological effects and catabolic metabolism of 3′ 5′-cyclic nucleotides and derivatives in rat adipose tissue and liver. Metabolism20, 63–77 (1971)
Butcher, R.W., Sutherland, E.W.: Adenosine 3′ 5′-phosphate in biologic materials. Purification and properties of cyclic 3′ 5′-nucleotide phosphodiesterase and use of this enzyme to characterize adenosine 3′ 5′-phosphate in human urine. J. biol. Chem.237, 1244–1250 (1962)
Carter, W.J., Younathan, E.S.: Studies on protection against the diabetogenic effect of alloxan by glucose. Proc. Soc. exp. Biol (N. Y.)109, 611–612 (1962)
Cooper, R.H., Ashcroft, S.J.H., Randle, P.J.: Concentration of adenosine 3′:5′-cyclic monophosphate in mouse pancreatic islets measured by a protein-binding radioassay. Biochem. J.134, 599–605 (1973)
Dean, D.M., Matthews, E.K.: The bioelectrical properties of pancreatic islet cells: effect of diabetogenic agents. Diabetologia8, 173–178 (1972)
Drummond, G.I., Powell, C.A.: Analogues of adenosine 3′ 5′-cyclic phosphate as activators of phosphorylase b kinase and as substrates for cyclic 3′ 5′-nucleotide phosphodiesterase. Mol. Pharmacol.6, 24–30 (1970)
Henion, W.F., Sutherland, E.W., Posternak, T.: Effects of derivatives of adenosine 3′ 5′-phosphate on liver slices in intact animals. Biochim. biophys. Acta (Amst.)148, 106–113 (1967)
Jarett, L., Smith. R.M.: Mode of action of N6-O2-dibutyryl cyclic 3′ 5′ AMP on fat cell metabolism. Diabetes23, 29–40 (1974)
Lacy, P.E., Walker, M.M., Fink, C.J.: Perifusion of isolated rat isletsin vitro: participation of the microtubular system in the biphasic release of insulin. Diabetes21, 987–998 (1972)
Lacy, P.E., Klein, N.J., Fink, C.J.: Effect of cytochalasin B on the biphasic release of insulin in perifused rat islets. Endocrinology92, 1458–1468 (1973)
Lacy, P.E., Kostianovsky, M.: Method for the isolation of intact islets of Langerhans from the rat pancreas. Diabetes16, 35–39 (1967)
Orci, L., Amherdt, M., Stauffacher, W., Like, A.A., Rouiller, C., Renold, A.E.: Structural changes in membranes of beta cells exposed to alloxan and streptozotocin demonstrated by freeze-etching. Diabetes21 (Suppl. 1), 326 (1972)
Rossini, A.A., Berger, M., Shadden, J., Cahill, G.F., Jr.: Beta cell protection to alloxan necrosis by anomers of D-glucose. Sience183, 424 (1974)
Sams, D.J. and Montague, W.: The role of adenosine 3′:5′-cyclic monophosphate in the regulation of insulin release. Biochem. J.129, 945–952 (1972)
Scheynius, A., Täljedal, I.-B.: On the mechanism of glucose protection against alloxan toxicity. Diabetologia7, 252–255 (1971)
Sutherland, E.W., Rall, T.W.: Fractionation and characterization of a cyclic adenosine ribonucleotide formed by tissue particles. J. biol. Chem.232, 1077–1091 (1958)
Tomita, T., Lacy, P.E., Matschinsky, F.M., McDaniel, M.L.: The effect of alloxan on insulin secretion in isolated rat islets perifusedin vitro. Diabetes23, 517–524 (1974)
Tomita, T., Lacy, P.E.: Interaction of alloxan and glucoseinduced insulin secretion of isolated rat islets perifusedin vitro. Diabetes21 (Suppl. 1), 326 (1972)
Watkins, D., Cooperstein, S.J., Lazarow, A.: Effect of alloxan on islet tissue permeability: protection and reversals by sugars. Amer. J. Physiol.224, 718–722 (1973)
Wright, P.H., Makulu, D.R., Vichick, D., Sussmann, K.E.: Insulin immunoassay by back-titration; some characteristics of the technique and the insulin precipitant action of alcohol. Diabetes20, 33–45 (1971)
Zawalich, W.S., Beidler, L.M.: Glucose and alloxan interactions in the pancreatic islets. Amer. J. Physiol.224, 963–966 (1973)
Author information
Authors and Affiliations
Additional information
This research was supported in part by NIH grants AM03373 and AM06181
Rights and permissions
About this article
Cite this article
Lacy, P.E., McDaniel, M.L., Fink, C.J. et al. Effect of methylxanthines on alloxan inhibition of insulin release. Diabetologia 11, 501–507 (1975). https://doi.org/10.1007/BF01222099
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01222099