Summary
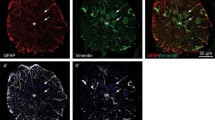
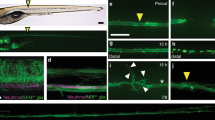
The nature and organization of the cellular substrate supporting axonal outgrowth during early regeneration of the spinal cord following transection and/or segment removal were examined inXenopus tadpoles. Longitudinal axonal compartments, formed by radial ependymal processes in unoperated spinal cords, were maintained within the rostral and caudal stumps throughout the early post-operative period. The first neuritic sprouts to appear near the cut ends of the cord were frequently associated with these processes. Between 5 and 7 days after transection either single or multiple cellular aggregates, which consisted primarily of ependyma, began occupying the lesion zone and were encompassed by numerous small fibres. Ependymal processes subsequently extended among the neuntes within the lesion gap and re-established longitudinal axonal channels and a surrounding glia limitans.
A concurrent outgrowth of fibres and ependyma from the rostral and caudal stumps was also initiated by 5–7 days following resection of the cord. Axons were seen further within the ablation gap at 10 and 12 days either coursing along the surfaces of the emerging ependyma or organized into small fascicles by the radial processes of these cells. Intermediate stages of axonal fasciculation by the underlying ependyma were also seen. In contrast with previous reports, axonal elongation beyond the cut ends of the cord did not consistently entail the formation of pre-existing ependymal channels. These observations provide additional evidence emphasizing the importance of the ependymal cell surface during axonal outgrowth in the regenerating spinal cord. Guided neuritic elongation, however, does not appear to be dependent upon the prior establishment of a specific type of cytoarchitecture.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bernstein, J. J. andBernstein, M. E. (1969) Ultrastructure of normal regeneration and loss of regenerative capacity following teflon blockage in goldfish spinal cord.Experimental Neurology 24, 538–57.
Butler, E. G. andWard, M. B. (1965) Reconstitution of the spinal cord following ablation in Urodele larvae.Journal of Experimental Zoology 160, 47–66.
Butler, E. G. andWard, M. G. (1967) Reconstitution of the spinal cord after ablation in adultTriturus. Developmental Biology 15, 464–86.
Brown, J. O. andMcCouch, G. p. (1947) Abortive regeneration of the transected spinal cord.Journal of Comparative Neurology 87, 131–7.
Clemente, C. D. (1964) Regeneration in the vertebrate central nervous system.International Review of Neurobiology 6, 257–301.
Clemente, C. D. andWindle, W. F. (1954) Regeneration of severed nerve fibres in the spinal cord of the adult cat.Journal of Comparative Neurology 101, 691–731.
Davidoff, L. M. andRansohoff, J. (1948) Absence of spinal cord regeneration in the cat.Journal of Neurophysiology 11, 9–11.
Egar, M., Simpson, S. B. andSinger, M. (1970) The growth and differentiation of the regenerating spinal cord of the lizard,Anolis carolinemis.Journal of Morphology 131, 131–52.
Egar, M. andSinger, M. (1972) The role of the ependyma in spinal cord regeneration in the urodele,Triturus.Experimental Neurology 37, 422–30.
Guth, L. (1975) History of central nervous system regeneration research.Experimental Neurology 8, 3–15.
Hooker, D. (1925) Studies on regeneration in the spinal cord. III. Re-establishment of anatomical and physiological continuity after transection in frog tadpoles.Journal of Comparative Neurology 38, 315–45.
Karnovsky, M. J. (1971) Use of ferrocyanide-reduced osmium tetroxide in electron microscopy. Abstracts,Eleventh Annual Meeting, American Society for Cell Biology, p. 146.
Michel, M. E. andReier, P. J. (1977) Degeneration and early neuronal outgrowth following spinal cord transection inXenopus laevis tadpoles.Neuroscience Abstracts 3, 504.
Murray, M. (1976) Regeneration of retinal axons into the goldfish optic tectum.Journal of Comparative Neurology 168, 175–96.
Nieuwkoop, P. D. andFaber, J. (1956)Normal Table of Xenopus laevis (Daudin). Amsterdam: North-Holland.
Nordlander, R. E. andSinger, M. (1978) The role of ependyma in regeneration of the spinal cord in urodele amphibian tail.Journal of Comparative Neurology 180, 349–74.
Pettegrew, R. K. andWindle, W. F. (1976). Factors in recovery from spinal cord injury.Experimental Neurology 53, 815–29.
Piatt, J. (1955) Regeneration in the central nervous system of amphibia. InRegeneration in the Central Nervous System (edited byWindle, W. F.), pp. 20–46. Springfield: Thomas.
Puchala, E., andWindle, W. F. (1977) The possibility of structural and functional restitution after spinal cord injury. A review.Experimental Neurology 55, 1–42.
Ramon Y Cajal, S. (1928)Degeneration and Regeneration of the Nervous System. Translated byR. M. May. London: Oxford University Press.
Reier, P. J. (1978) An ultrastructural analysis of early axonal outgrowth during optic nerve regeneration inXenopus laevis tadpoles.Anatomical Record 190, 159 (Abstract).
Reier, P. J. (1979) Penetration of grafted astrocytic scars by regenerating optic nerve axons inXenopus tadpoles.Brain Research 164, 61–8.
Reier, P. J. andWebster, H. deF. (1974) Regeneration and remyelination ofXenopus tadpole optic nerve fibres following transection or crush.Journal of Neurocytology 3, 591–618.
Rovainen, C. M. (1976) Regeneration of Müller and Mauthner axons after spinal transection in larval lampreys.Journal of Comparative Neurology 168, 545–54.
Selzer, M. E. (1978) Mechanisms of functional recovery and regeneration after spinal cord transection in larval sea lamprey.Journal of Physiology 279, 395–408.
Sims, R. T. (1962) Transection of the spinal cord in developingXenopus laevis.Journal of Embryology and Experimental Morphology 10, 115–26.
Simpson, S. B. (1968) Morphology of the regenerated spinal cord in the lizard,Anolis carolinensis.Journal of Comparative Neurology 134, 193–210.
Turner, J. E. andSinger, M. (1974) The ultrastructure of regeneration in the severed new optic nerve.Journal of Experimental Zoology 190, 249–68.
Varon, S. S. andBunge, R. P. (1978) Trophic mechanisms in the peripheral nervous system.Annual Review of Neuroscience 1, 327–61.
Weiss, P. (1955) Neurogenesis. InAnalysis of Development (edited byWillier, B. H., Weiss, P. andHamburger, V.), pp. 345–401. Philadelphia: Saunders.
Windle, W. F. (1956) Regeneration of axons in the vertebrate central nervous system.Physiological Reviews 36, 427–40.
Windle, W. F. andChambers, W. W. (1950) Spinal cord regeneration associated with a cellular reaction induced by administration of a purified bacterial pyrogen.Fifth International Anatomical Congress, Oxford, p. 196.
Young, J. Z. (1942) The functional repair of nervous tissue.Physiological Reviews 22, 318–74.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Michel, M.E., Reier, P.J. Axonal-ependymal associations during early regeneration of the transected spinal cord inXenopus laevis tadpoles. J Neurocytol 8, 529–548 (1979). https://doi.org/10.1007/BF01208508
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01208508