Abstract
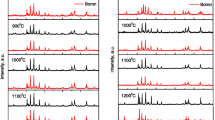
The formation mechanism, morphology, and magnetic properties of anisotropic BaFe12O19 powders prepared by the 0.8 Na2SO4-0.2 K2SO4 molten salt synthesis have been investigated. The reaction can be completed at temperatures lower than 845 °C which is the melting point of the 0.8 Na2SO4-0.2 K2SO4 salt mixture, due to the presence of the low melting phase related to the formation of BaSO4 and Na6CO3(SO4)2. Even at temperatures lower than 845 °C, with the aid of the low melting phase, the morphology of ferrites was thin platelets. Magnetic properties such as coercivity, saturation magnetization, and remanent force are comparable to those found in other processing techniques. The orientation degree of the sintered body was very high due to anisotropic morphology; thus powders from molten salt synthesis are suitable for grain oriented ceramics.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
K. Haneda, C. Miyakawa andH. Kojima,J. Amer. Ceram. Soc. 57 (1974) 354.
W. Ross,ibid. 63 (1980) 6011.
C. Mee andJ. Jeschke,J. Appl. Phys. 34 (1963) 1271.
B. Beer andG. Planar,Brit. Commun. Electron. 5 (1958) 939.
A. Srivastava, P. Singh andM. Gupta,J. Mater. Sci. 22 (1987) 1489.
K. Higuchi, S. Naka andS. Hirano,J. Ceram. Soc. Jpn. 95 (1987) 186.
Idem, Adv. Ceram. Mater. 1 (1986) 104.
Idem, ibid. 3 (1988) 278.
Idem, ibid. 2 (1987) 65.
M. Kiyama,Bull. Chem. Soc. Jpn. 49 (1976) 1855.
B. Shirk andW. Buessem,J. Amer. Ceram. Soc. 53 (1970) 192.
R. Arendt,J. Solid State Chem. 8 (1973) 339.
D. G. Wickham, in “Proceedings of the 1st International Conference on Ferrites”, Kyoto, July 1970, edited by Y. Hoshino, S. Iida and M. Sugimoto (University Park Press, Baltimore, 1971) p. 105.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoon, K.H., Lee, D.H., Jung, H.J. et al. Molten salt synthesis of anisotropic BaFe12O19 powders. J Mater Sci 27, 2941–2945 (1992). https://doi.org/10.1007/BF01154103
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01154103