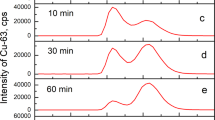
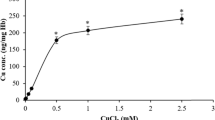
Summary
Upon incubation of peripheral leucocytes with copper sulphate a dramatic cellular copper uptake reaching levels of 25–50-fold compared to that of the natural copper content was measured. The orange-red fluorescence of the copper-treated white blood cells was assigned to the formation of Cu(I)-thiolate clusters in Cu(I)-thionein. A protein of 6–8 kDa was isolated from homogenized bovine leucocytes and characterized by its electronic absorption and amino acid composition to be identical to the above Cu(I)-thionein. More than 70% of the intracellular copper was attributed to this protein in its monomeric and polymeric form. Cu-thionein formation was more pronounced in monocytes than in granulocytes. As most intriguing phenomenon, the release of this Cu-thionein from leucocytes, was also noticed. The occurrence of Cu-thionein in leucocytes and the excretion of the intact Cu(I)-thiolate protein is of considerable interest with respect to the observed elevated copper levels in white blood cells and plasma during tumor malignancies and inflammatory processes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anglin JH, Batten WH, Raz AI, Sayre RM (1971) Fluorescence of Cu, Au, and Ag mercaptides. Photochem Photobiol 13:279–281
Beltramini M, Lerch K (1981) Luminescence properties of Neurospora copper metallothionein. FEBS Lett 127:201–203
Carpentieri U, Myers J, Thorpe L, Daeschner CW, Haggard ME (1986) Copper, zinc and iron in normal and leukemic lymphocytes from children. Cancer Res 46:981–984
Deuschle U, Weser U (1985) Copper and inflammation. Prog Clin Biochem 2:99–130
Dioguardi N, Agostini A, Fiorelli G (1963) Characterization of lactic dehydrogenase of normal human granulocytes. J Lab Clin Med 61:713–723
Felix K, Hartmann H-J, Weser U (1989) Cu(I)-thionein release from copper-loaded yeast cells. Biol Metals 2:50–54
Gutteridge JMC (1985) Inhibition of the Fenton reaction by the protein caeruloplasmin and other copper complexes. Assessment of ferroxidase and radical scavening activities. Chem-Biol Interact 56:113–120
Hartmann H-J, Weser U (1977) Copper-thionein from fetal bovine liver. Biochim Biophys Acta 491:211–222
Hartmann H-J, Gärtner A, Weser U (1985) Copper-dependent control of the enzymic and phagocyte induced degradation of some biopolymers, a possible link to systemic inflammation. Clin Chim Act 152:95–103
Hartmann H-J, Morpurgo L, Schechinger T, Desideri A, Rotilio G, Weser U (1987) Functional aspects of copper-thiolate centres in copper thionein. In: Bannister JV, Michelson AM (eds) Life chemistry reports. Harwood Academic Publishers, London, pp 305–318
Holtzman NA, Gaumnitz BM (1970) Identificaton of an apoceruloplasmin-like substance in the plasma of copper-deficient rats. J Biol Chem 245:2350–2353
Kataoka M, Tavassoli M (1985) Identification of ceruloplasmin receptors on the surface of human blood monocytes, granulocytes, and lymphocytes. Exp Hematol 13:806–810
Koizumi S, Sone T, Kimura M, Otsuka F, Ohsawa M (1987) Metallothioneins of monocytes and lymphocytes. In: Kägi JHR, Kojima Y (eds) Metallothionein II. Birkhäuser, Basel, pp 519–523
Peavy DL, Fairchild EJ (1987) Induction of metallothionein synthesis in human peripheral blood leukocytes. Environ Res 42:377–385
Porter H (1974) The particulate half-cystine-rich copper protein of newborn liver. Relationship of metallothionein and subcellular localization in non-mitochondrial particles possibly representing heavy lysosomes. Biochem Biophys Res Commun 56:661–668
Richter A, Weser U (1988) Kinetics of the H2O2-dependent cleavage of Cu-thiolate centres in yeast Cu8-thionein. Inorg Chim Acta 151:145–148
Riordan JR, Madapallimattam G (1979) Low molecular weight copper-binding proteins in cultured human cells. In: Kägi JHR, Nordberg M (eds) Metallothionein. Birkhäuser, Basel, pp 353–361
Rupp H, Weser U (1974) Conversion of metallothionein into Cu-thionein, the possible low molecular weight form of neonatal hepatic mitochondrocuprein. FEBS Lett 47:293–297
Schechinger T, Hartmann H-J, Weser U (1986) Copper transport from Cu(I)-thionein into apo-caerulosplasmin mediated by activated leukocytes. Biochem J 240:281–283
Weser U, Hartmann H-J (1984) Copper-thiolate proteins (metalloproteins). In: Lontie R (ed) Copper proteins and copper enzymes. CRC Press, Boca Raton FL, pp 151–173
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hartmann, HJ., Schechinger, T. & Weser, U. Copper-thionein in leucocytes. Biol Metals 2, 40–44 (1989). https://doi.org/10.1007/BF01116200
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01116200