Abstract
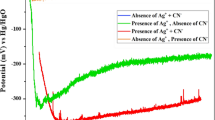
A systematic electrochemical study of pyrite in H2SO4 solutions containing dissolved silver was undertaken to gain more information about the transfer of silver ions to pyrite and their role in enhancing the direct oxidation of pyrite. The results of cyclic voltammetry experiments provide additional evidence of the formation of metallic silver on the FeS2 surface under open-circuit conditions. A pyrite electrode held at the open-circuit potential for 2 h in the presence of 10−3 m Ag+ exhibits a large and sharp anodic peak at about 0.7V. The current associated with this peak is the result of the dissolution of metallic silver deposited during the initial conditioning period. There is no evidence of silver deposition without preconditioning until the potential drops below about 0.6V for Ag+ concentrations ranging from 10−4 to 10−2 m. However, subsequent silver deposition appears to be very sensitive to the dissolved silver concentration in this range. There is also evidence that the state of the pyrite surface has a pronounced influence on its interaction with silver ions. Agitation has also been found to have a significant effect on the electrochemistry of the Ag−FeS2 system.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
R. S. Kunter, J. R. Turney and R. D. Lear, Presentation at International Precious Metals Symposium, TMS-AIME, Los Angeles, California, 26 February–1 March, 1984.
R. M. G. S. Berezowsky and D. R. Weir,Min. Metall. Process. 1 (1984) 1.
R. W. Lawrence and A. Bruynesteyn,CIM Bull 76 (1983) 107.
R. P. Hackl, F. Wright and A. Bruynesteyn, Presentation at Biotechnology for Gold Industry Symposium, CIM, Toronto, Ontario, 17–20 August 1986.
P. B. Munoz, J. D. Miller and M. E. Wadsworth,Metall. Trans. 10B (1979) 149.
G. J. Snell and M. C. Sze,EMJ 178 (1977) 100.
F. E. Pawlek, in ‘Extractive Metallurgy of Copper’ edited by J. C. Yannopoulos and J. C. Agarwal, AIME, New York (1976) Vol. II, Ch. 35.
J. D. Miller and H. Q. Portillo,Dev. Miner. Process 2 (1981) 851.
R. Y. Wan, J. D. Miller, J. Foley and S. Pons, ‘Electrochemistry in Mineral and Metal Processing’, Electrochem. Soc. (1984) p. 205.
D. W. Price and G. W. Warren,Hydrometallurgy 15 (1986) 303.
D. W. Price, G. W. Warren and B. Drouven,J. Appl. Electrochem. 16 (1986) 719.
J. B. Hiskey, P. P. Phule and M. D. Pritzker,Metall. Trans.,18B (1987) 641.
T. Biegler and D. A. Swift,Electrochim. Acta 24 (1979) 415.
R. E. Meyer,J. Electroanal. Chem. 101 (1979) 59.
I. C. Hamilton and R. WoodsJ. Electroanal. Chem. 118 (1981) 327.
T. Biegler,J. Electroanal. Chem. 70 (1976) 265.
L. K. Bailey and E. Peters,Can. Met. Quart. 15 (1976) 333.
E. Peters and H. Majima,Can. Met. Quart. 7 (1968) 111.
S. Fletcher et al.,J. Electroanal. Chem. 159 (1983) 267.
S. Fletcher,J. Electroanal. Chem. 118 (1981) 419.
J. Keizer and D. Scherson,J. Phys. Chem. 84 (1980) 2025.
R. DeLevie,J. Electroanal. Chem. 25 (1970) 257.
M. Korolczuk and J. Matysik,J. Electroanal. Chem. 193 (1985) 277.
S. Whiteway, D. MacLennan and C. CoffinJ. Chem. Phys. 18 (1950) 473.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hiskey, J.B., Pritzker, M.D. Electrochemical behavior of pyrite in sulfuric acid solutions containing silver ions. J Appl Electrochem 18, 484–490 (1988). https://doi.org/10.1007/BF01093767
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01093767