Abstract
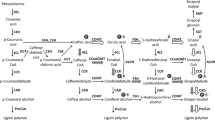
Three different forms of hydroxycinnamate: CoA ligase (EC 6.2.1-) have been separated by chromatofocusing from poplar stems. These three forms exhibit different substrate specificities and tissue distribution. A correlation was established between the monomeric composition of lignins isolated from xylem and sclerenchyma and the pattern of hydroxycinnamate: CoA ligase isoforms in these tissues. The results obtained indicate that, in poplar, the hydroxycinnamate: CoA ligase isoenzymes could play an important role in the control of the monomeric composition of lignins.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem.72, 248–254
Grand, C., Ranjeva, R. (1979) La lignification chez le Peuplier. III. Variations du niveau d'activité d'enzymes impliquées dans la biosynthèse des monomères en fonction du degré de lignification et de la nature des tissus. Physiol. Vég.17, 433–444
Grand, C., Boudet, A.M., Ranjeva, R. (1982) Natural variations and controlled changes in lignification process. Holzforschung36, 217–223
Gross, G.G., Mansell, R.L., Zenk, M.H. (1975) Hydroxycinnamate: coenzyme A ligase from lignifying tissues of higher plants. Biochem. Physiol. Pflanz.168, 41–51
Heinzmann, U., Seitz, U., Seitz, U. (1977) Purification and substrate specificities of hydroxycinnamate: CoA ligase from anthocyanin-containing and anthocyanin-free carrot cells. Planta135, 313–318
Knobloch, K.H., Hahlbrock, K. (1975) Isoenzymes ofp-coumarate: CoA ligase from cell suspension cultures ofGlycine max. Eur. J. Biochem.52, 311–320
Knobloch, K.H., Hahlbrock, K. (1977) 4-coumarate: CoA ligase from cell suspension cultures ofPetroselinum hortense Hoffm. Arch. Biochem. Biophys.184, 237–248
Knogge, W., Beulen, C., Weissenböck, G. (1981) Distribution of phenylalanine ammonialyase and 4-coumarate: CoA ligase in oat primary leaf tissues. Z. Naturforsch.36c, 389–395
Kuroda, H., Shimada, M., Higuchi, T. (1975) Purification and properties of O-methyl transferase involved in the biosynthesis of Gymnosperms lignin. Phytochemistry14, 1759–1763
Kutsuki, H., Shimada, M., Higuchi, T. (1982a) Regulatory role of cinnamyl alcohol dehydrogenase in the formation of guaïacyl and syringyl lignins. Phytochemistry21, 19–23
Kutsuki, H., Shimada, M., Higuchi, T. (1982b) Distribution and roles ofp-hydroxycinnamate: CoA ligase in lignin biosynthesis. Phytochemistry21, 267–271
Lüderitz, T., Grisebach, H. (1981) Enzymic synthesis of lignin precursors. Comparison of cinnamoyl-CoA reductase and cinnamyl alcohol: NADP+ dehydrogenase from Spruce (Pica abies L.) and Soybean (Glycine max L.). Eur. J. Biochem.119, 115–124
Lüderitz, T., Schatz, G., Grisebach, H. (1982) Enzymic synthesis of lignin precursors. Purification and properties of 4-coumarate: CoA ligase from cambial sap of Spruce (Picea abies L.). Eur. J. Biochem.123, 583–586
Ranjeva, R., Boudet, A.M., Faggion, R. (1976) Phenolic metabolism in petunia tissues. IV. Properties ofp-coumarate: coenzyme A ligase isoenzymes. Biochimie58, 1255–1262
Shimada, M., Kuroda, H., Higuchi, T. (1973) Evidence for the formation of methoxyl groups of ferulic and sinapic acids inBambusa by the same O-methyl-transferase. Phytochemistry12, 2873–2875
Stöckigt, J., Zenk, M.H. (1975) Chemical syntheses and properties of hydroxycinnamoyl-CoA derivatives. Z. Naturforsch.30c, 352–356
Wallis, P.J., Rhodes, M.J.C. (1977) Multiple forms of hydroxycinnamate: CoA ligase in etiolated pea seedlings. Phytochemistry16, 1891–1894
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grand, C., Boudet, A. & Boudet, A.M. Isoenzymes of hydroxycinnamate: CoA ligase from poplar stems properties and tissue distribution. Planta 158, 225–229 (1983). https://doi.org/10.1007/BF01075258
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01075258