Abstract
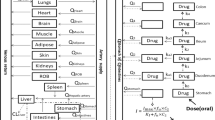
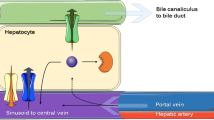
The potential influence of alterations in several physiologic processes (hepatocellular egress, biliary excretion, gastrointestinal transit) and biotransformation steps (oxidative metabolism, glucuronidation) on the disposition of agents subject to significant enterohepatic recirculation (ER) via the glucuronide conjugate was examined in a series of simulation experiments. The model of ER developed was based upon the disposition of valproic acid (VPA) and valproate glucuronide (VPA-G) in the rat. The systemic disposition of VPA was simulated following changes in several processes contributing to (or competing with) ER: hepatic oxidative metabolism, hepatic glucuronidation, sinusoidal egress of glucuronide conjugate, canalicular egress of glucuronide conjugate, and gastrointestinal transit. Changes in the formation clearance of VPA-G resulted in a less than proportional change in systemic clearance of VPA, whereas changes in oxidative metabolism led to a greater than proportional change in systemic clearance. Furthermore, alterations in hepatocellular egress of VPA-G affected the disposition of the parent compound, suggesting that drug interactions or disease state effects on metabolite transport may be misinterpreted as effects at the level of metabolite formation. Analytical methods are proposed to recover the intrinsic kinetic parameters (formation clearances of metabolites, renal clearance of parent, volume of distribution) in the presence of ER from the systemic disposition of the parent alone.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B. E. Dahlstrom and L. K. Paalzow. Pharmacokinetic interpretation of the enterohepatic recirculation and first-pass elimination of morphine in the rat.J. Pharmacokin. Biopharm. 6:505–519 (1978).
J. H. Lin, K. C. Yen, and D. E. Duggan. Effect of enterohepatic circulation on the pharmacokinetics of diflunisal in rats.Drug Metab. Dispos. 13:321–326 (1985).
R. J. Herman, J. D. Van Pham, and C. B. N. Szakacs. Disposition of lorazepam in human beings: Enterohepatic recirculation and first-pass effect.Clin. Pharmacol. Ther. 46:18–25 (1989).
F. L. S. Tse, F. Ballard, and J. Skinn. Estimating the fraction reabsorbed for drugs undergoing enterohepatic circulation.J. Pharmacokin. Biopharm. 10:455–461 (1982).
R. G. Dickinson, R. C. Harland, A. M. Ilias, R. M. Rodgers, S. N. Kaufman, R. K. Lynn, and N. Gerber. Disposition of valproic acid in the rat: Dose-dependent metabolism, distribution, enterohepatic recirculation, and choleretic effect.J. Pharmacol. Exp. Ther. 211:583–595 (1979).
K. Singh, J. M. Orr, and F. S. Abbott. Pharmacokinetics and enterohepatic circulation of 2-n-propyl-4-pentenoic acid in the rat.Drug Metab. Dispos. 16:848–852 (1988).
O. J. McAnena, M. Rossi, B. M. Mehta, and J. M. Daly. Alteration of methotrexate metabolism in rats by administration of an elemental liquid diet. I. Changes in drug enterohepatic circulation.Cancer 59:31–37 (1987).
K. C. Kwan, G. O. Breault, E. R. Umbenhauer, F. G. McMahon, and D. E. Duggan. Kinetics of indomethacin absorption, elimination, and enterohepatic circulation in man.J. Pharmacokin. Biopharm. 4:255–280 (1976).
T. R. D. Elmhirst, J. K. Chipman, O. Ribeiro, P. C. Hirom, and P. Millburn. Metabolism and enterohepatic circulation of benzo(a)pyrene-4,5-epoxide in the rat.Xenobiotica 15:899–906 (1985).
H. Kurebayashi, A. Tanaka, and T. Yamaha. Metabolism and disposition of the flame retardant plasticizer, tri-p-cresyl phosphate, in the rat.Toxicol. Appl. Pharmacol. 77:395–404 (1985).
D. E. Duggan, K. F. Hooke, R. M. Noll, and K. C. Kwan. Enterohepatic circulation of indomethacin and its role in intestinal irritation.Biochem. Pharmacol. 25:1749–1754 (1975).
R. Gugler and G. E. von Unruh. Clinical pharmacokinetics of valproic acid.Clin. Pharmacokin. 5:67–83 (1980).
F. Schobben and E. van der Kleijn. Valproate biotransformation. In D. M. Woodbury, J. K. Penry, and C. E. Pippinger (eds.),Antiepileptic Drugs, Raven, New York, 1982, pp. 567–578.
A. Rescigno and G. Segre.Drug and Tracer Kinetics, Blaisdell, Waltham, MA, 1966.
J. Barre, J. M. Chamouard, G. Houin, and J. P. Tillement. Equilibrium dialysis, ultrafiltration, and ultracentrifugation compared for determining the plasma-protein-binding characteristics of valproic acid.Clin. Chem,31:60–64 (1985).
W. Loscher. Rapid determination of valproate sodium in serum by gas-liquid chromatography.Epilepsia 18:782–787 (1978).
W. Loscher and H. Esenwein. Pharmacokinetics of sodium valproate in dog and mouse.Arzneim. Forsch. 28:782–787 (1978).
H. Akaike. An information criterion.Math. Sci. 14:5–9 (1976).
M. Gibaldi and D. Perrier.Pharmacokinetics, 2nd ed. Marcel Dekker, New York, 1985.
U. Bredberg and L. Paaizow. Pharmacokinetics of methylergometrine in the rat: Evidence for enterohepatic recirculation by a linked-rat model.Pharm. Res. 7:14–20 (1990).
G. M. Pollack. Pharmacokinetic factors contributing to the nonlinear disposition of valproic acid in the rat.Pharm. Res. 3:145 (1986).
G. D. Anderson, A. Acheampong, A. J. Wilensky, and R. H. Levy. Effect of valproate (VPA) dose on formation of hepatotoxic metabolites.Clin. Pharmacol. Ther. 47:130 (1990).
P. veng Pedersen and R. Miller. Pharmacokinetics of doxycycline reabsorption.J. Pharm. Sci. 69:204–207 (1980).
J.-L. Steimer, Y. Plusquellec, A. Guillaume, and J.-F. Boisvieux. A time-lag mode for Pharmacokinetics of drugs subject to enterohepatic circulation.J. Pharm. Sci. 71:297–302 (1982).
Y. Plusquellec and L. Bousquet. Time-delay for two-compartment models used for study of enterohepatic circulation of drugs.IEEE Trans. Biomed. Eng. 6:469–472 (1984).
T. A. Shepard, D. J. Gannaway, and G. F. Lockwood. Accumulation and time to steady state for drugs subject to enterohepatic cycling: A simulation study.J. Pharm. Sci. 74:1331–1333 (1985).
W. A. Colburn. Pharmacokinetic analysis of concentration-time data obtained following administration of drugs that are recycled in the bile.J. Pharm. Sci. 73:313–317 (1984).
J. G. Wagner, A. R. DiSanto, W. R. Gillespie, and K. S. Albert. Reversible metabolism and pharmacokinetics: Application to prednisone-prednisolone.Res. Comm. Chem. Path. Pharmacol. 32:387–405 (1981).
S. Hwang, K. C. Kwan, and K. S. Albert. A linear model of reversible metabolsim and its application to bioavailability assessment.J. Pharmacokin. Biopharm. 9:693–709 (1981).
W. F. Ebling and W. J. Jusko. The determination of essential clearance, volume, and residence time parameters of recirculating metabolic systems: The reversible metabolism of methylprednisolone and methylprednisone in rabbits.J. Pharmacokin. Biopharm. 14:557–599 (1986).
H. Cheng and W. J. Jusko. Mean residence times of multicompartmental drugs undergoing reversible metabolism.Pharm. Res. 7:103–107 (1990).
R. G. Dickinson, R. M. Kluck, M. J. Eadie, and W. D. Hooper. Disposition ofβ- glucuronidase-resistant “glucuronides” of valproic acid after intrabiliary administration in the rat: Intact absorption, fecal excretion, and intestinal hydrolysis.J. Pharmacol. Exp. Ther. 233:214–221 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pollack, G.M., Brouwer, K.L.R. Physiologic and metabolic influences on enterohepatic recirculation: Simulations based upon the disposition of valproic acid in the rat. Journal of Pharmacokinetics and Biopharmaceutics 19, 189–225 (1991). https://doi.org/10.1007/BF01073869
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01073869