Abstract
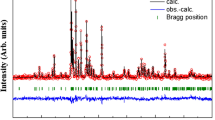
A natural sample of ankerite has been characterized by chemical analysis, X-ray diffraction and differential thermal analysis. The composition was found to be (Ca1.11Mg0.50Fe0.33Mn0.09) [Co3]1.99.57Fe Mössbauer effect measurements were performed at temperatures between 4.2 and 400 K. At low temperatures (T < 25 K) relaxation effects are clearly dominant. The temperature dependence of the center shift is remarkably well reproduced by a model based on the Debije approximation of the lattice vibrations. In contrast, the temperature dependence of the quadrupole splitting cannot be described by any reasonable crystal field model. It is argued that an orbit-lattice coupling might explain the observed quadrupole splittings. A spectrum recorded in an applied field of 6 T reveals a positive electric field gradient from which an orbital doublet ground state is concluded. Highly anisotropic field reductions are derived but cannot be quantitatively explained due to a lack of knowledge concerning the magnetic structure of ankerite. The line widths decrease significantly with increasing temperature which is only partly due to the decreasing Mössbauer fraction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bancroft GM (1973) Mössbauer spectroscopy. An introduction for inorganic chemists and geochemists. McGraw Hill, London, pp 110–154
Blume M (1968) Stochastic theory of line shape: generalization of the Kubo-Anderson model. Phys Rev 174:351–358
Bragg L (1965) Crystal structures of minerals, Vol 4. G Bell and Sons Ltd., London, p 127
Cohen M (1935) Precision lattice constants from X-ray powder photographs. Rev Sci Instrum 6:68–74
Cole RD, Liu J, Smith GV, Hinckley CC, Saporoschenko M (1978) Iron partitioning in oil shale of the Green River Formation, Colorado: a preliminary Mössbauer study. Fuel 57:514–520
De Grave E, Bowen LH, Hedges SW (1982) Mössbauer spectroscopy with a microprocessor: a versatile software package. Nucl Instrum Methods 200:303–310
De Grave E, Vanleerberghe R, Verdonck L, De Geyter G (1984) Mössbauer and infrared spectroscopic studies of Belgian chloritoids. Phys Chem Minerals 11:85–94
Dockum BW, Reiff WM (1979) A high field Mössbauer study of a six coordinate iron II dithiocarbamate complex and comparison of the Fe(II)S6, Fe(III)S6 and Fe(IV)S6 chromophores in tris(diethyldithiocarbamate) complexation. Chem Phys Letters 63:32–36
Ericsson T, Wäppling R (1976) Texture effects in 3/2–1/2 Mössbauer spectra. J Phys Colloq 37:C6-719–C6-723
Frauenfelder H, Nagle DE, Taylor RD, Cochran DRF, Visscher WM (1962) Elliptical polarization of Fe57 gamma rays. Phys Rev 126:1065–1075
Gallagher PK, West KW, Warne SSJ (1981) Use of the Mössbauer effect to study the thermal decomposition of siderite. Thermochim Acta 50:41–47
Hang Nam OK (1969) Relaxation effects in antiferromagnetic ferrous carbonate. Phys Rev 185:472–476
Hanscom R (1980) The structure of triclinic chloritoid and chloritoid polymorphism. Am Mineral 65:534–539
Heberle J (1968) Linewidth of Mössbauer absorption. Nucl Instrum Methods 58:90–92
Heberle J (1971) The Debije integrals, the thermal shift and the Mössbauer fraction. In: Gruverman IJ (ed) Mössbauer effect methodology, Vol 7. Plenum Press, New York London, pp 299–308
Howie RA, Broadhurst FM (1958) X-ray data for dolomite and ankerite. Am Mineral 43:1210–1214
Ingalls R (1964) Electric field gradient tensor in ferrous compounds. Phys Rev 133:A787-A795
Johnson CE (1967) Hyperfine interactions in ferrous fluosilicate. Proc Phys Soc 92:748–757
Lefelhocz JF, Friedel RA, Kohman TP (1967) Mössbauer spectroscopy of iron in coal. Geochim Cosmochim Acta 31:2261–2273
Melchior DC, Wildeman TR, Williamson DL (1982) Mössbauer investigation of the transformations of the iron minerals in oil shale during retorting. Fuel 61:516–522
Montano PA (1981) Application of Mössbauer spectroscopy to coal characterization and utilization. In: Stevens JG, Shenoy GK (eds) Mössbauer spectroscopy and its chemical applications. American Chemical Society, Washington DC, pp 135–175
Mørup S, Lindgreen H (1982) Applications of Mössbauer spectroscopy in oil prospecting. In: Proceedings of the international conference on the applications of the Mössbauer effect. Indian National Science Academy, New Delhi, pp 290–292
Musić S, Nagy-Czakó I, Vértes A, HadĂija O (1980) Mössbauer effect study of siderite and limonite ores. Bull Soc Chim Beograd 45:541–547
Nagy DL, Dézsi I, Gonser U (1975) Mössbauer studies of FeCO3. Neues Jahrb Mineral Monatsh 1975:101–114
O'Connor DA (1963) The effect of line broadening of Mössbauer resonant sources and absorbers on the resonance absorption. Nucl Instrum Methods 21:318–322
Pound RV, Rebka GA Jr (1960) Variation with temperature of the energy of recoil-free gamma rays from solids. Phys Rev Letters 4:274–277
Povarennykh AS (1972) Crystal chemical classification of minerals, Vol 2. Plenum Press, New York London, pp 607–627
Pratt JH (1894) On the determination of ferrous iron in silicates. Am J Sci 48:149–151
Price DC (1978) Vibronic effects in Mössbauer spectra: the57Fe quadrupole splitting in FeCO3. Aust J Phys 31:397–420
Price DC, Srivastava KKP (1976) Paramagnetic hyperfine structure and electronic relaxation of Fe2+ in CaCO3 and CdCO3. J Phys Colloq 37:C6-123–C6-127
Shiley RH, Cluff RM, Dickerson DR, Hinckley CC, Smith GV, Twardowska H, Saporoschenko M (1981) Correlation of natural gas content to iron species in the New Albany shale group. Fuel 60:732–738
Smythe JA, Dunham KC (1947) Ankerites and chalybites from the northern Pennine orefield and the northeast coalfield. Min Mag 28:53–74
Spiering H, Zimmerman R, Ritter G (1974) Investigation of hyperfine interaction and structure in FeSiF6·6H2O by Mössbauer measurements. Phys Status Solidi (b) 62:123–133
Srivastava KKP (1983) Mössbauer quadrupole splitting of Fe2+ in carbonates. J Phys C: Solid State Phys 16:L1137-L1139
Visser JW (1969) Fully automatic program for finding the unit cell from powder data. Appl Crystallogr 2:89–95
Vogel AI (1962) Textbook of quantitative inorganic analysis. Long-mans, Green and Co. Ltd. London, pp 584–586
Varret F (1976) Mössbauer spectra of paramagnetic powders under applied field: Fe3+ in fluosilicates. J Phys Chem Solids 37:265–271
Warne SSJ, Morgan DJ, Milodowski AE (1981) Thermal analysis studies of the dolomite, ferroan dolomite, ankerite series. Part 1. Iron content recognition and determination by variable atmosphere DTA. Thermochim Acta 50:105–111
Wasastjerna JA (1924) The crystal structure of dolomite. Soc Scient Fennica Comm Phys — Math II, No 14
Williamson DL, Melchior DC (1980) Changes in iron minerals during oil-shale retorting. In Gary JH (ed) Proceedings of the 13th oil shale symposium. Colorado School of Mines Press, Golden, Colorado, pp 337–350
Author information
Authors and Affiliations
Additional information
Research Associate with the National Fund for Scientific Research (Belgium)
Rights and permissions
About this article
Cite this article
De Grave, E., Vochten, R. An57Fe mössbauer effect study of ankerite. Phys Chem Minerals 12, 108–113 (1985). https://doi.org/10.1007/BF01046835
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01046835