Summary
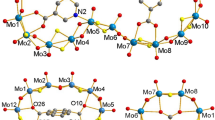
The complexes [PPh4][MoO{S(CH2)nS}2], n=2 (1) n=3 (3), were synthesised from the MoVI dioxoprecursor complex [MoO2(butane-2,3-diolato)2] and the appropriate thiol in the presence of base. Complex (3) exhibits a square pyramidal geometry. Crystal Data: P21,0, Z=4,a=9.612(2),b=21.202(4),c=15.462(3) Å, β=103.06(1)°, V=3043.3Å3, F(000)=1364.0, 3687 reflections collected, and 1957 used in solution with R=0.058. [MoO(SCH2CH2CH2S)2]− reacts with hydrazoic acid in methanol to produce [Mo2O2(μ-N3)-(SCH2CH2CH2S)3] (5), crystal data:\(P_{2_{1} 2_{1} 2_{1} } \) a=9.398(3),b=14.650(2),c=27.42(2) (Å), α=β=γ=90°, Z=4 V=3775.8Å3, F(000)=1872.0, 2563 reflections collected and 1244 were used in the solution with R=0.058. The complex is an asymmetric dimer with a triple bridge comprising two dithiolate sulphurs and a nitrogen of the azide ligand. Convolution analysis of the cyclic voltammetric response of complex (1) confirmed a diffusion controlled one electron reversible reduction process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. Hanson, A. A. Brunette, A. C. MacDonald, K. S. Murray and A. G. Wedd,J. Am. Chem. Soc.,103, 1953 (1981).
I. W. Boyd, I. G. Dance, K. S. Murray and A. G. Wedd,Aust. J. Chem.,31, 2423 (1978).
R. J. Burt, G. J. Leigh, J. R. Dilworth and J. A. Zubieta,J. Chem. Soc. Dalton Trans., 2295 (1982).
S. R. Ellis, O. Collison, C. D. Garner and W. Clegg,J. Chem. Soc. Chem. Commun., 1483 (1986).
P. T. Bishop, J. R. Dilworth, J. Hutchinson and J. A. Zubieta,J. Chem. Chem. Commun., 1053 (1982).
I. W. Boyd, I. G. Dance, A. E. Landors and A. G. Wedd,Inorg. Chem.,18, 1875 (1979).
C. D. Garner, L. H. Hill, F. E. Mabbs, F. E. McFadden and A. T. McPhail,J. Chem. Soc. Dalton Trans., 853 (1974).
P. M. Boormanm, C. D. Garner, F. E. Mabbs and T. J. King,J. Chem. Soc. Chem. Commun., 663 (1974).
P. J. Blower, J. R. Dilworth, J. Hutchinson and J. A. Zubieta,J. Chem. Soc. (Dalton), 1339 (1986).
Z. Dori and R. F. Ziolo,Chem. Rev.,97, 1616 (1975).
J. R. Dilworth, B. D. Neaves, P. Dalstrom, J. Hyde and J. A. Zubieta,Trans. Met. Chem.,7, 257 (1982).
G. Benzey, J. H. Enemark, J. I. Gelder and K. Yamanouchi, in P. C. Metchell and J. Seaman, (Eds.), Proceedings, Second International Conference on Chemistry and Uses of Molybdenum, Climax, Michigan (1977) p. 50.
R. S. Nicolson and I. Shain,Anal. Chem.,36, 706 (1964).
A. Blagg, S. W. Carr, G. R. Cooper, I. Dobson, J. Bernard Gill, D. C. Goodall, B. L. Shaw, N. Taylor and T. Boddington,J. Chem. Soc. Dalton Trans., 1213 (1985) and references therein.
F. E. Woodward, R. O. Goodin and P. J. Kinlen,Anal. Chem.,56, 1920 (1984).
J. C. Imbeaux and J. M. Savéant,Electroanal. Chem., andInterfacial Electrochem.,44, 169 (1973).
E. G. and CONDECON 300(TM) Version 2.0.
R. J. Butcher, H. K. J. Powell and S. M. Yong,J. Chem. Soc. Dalton Trans.,356 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bishop, P.T., Dilworth, J.R., Hutchinson, J.P. et al. Syntheses and x-ray crystal structures of [MoO(SCH2CH2-CH2S)2]− and [Mo2O2(μ-N3)(SCH2CH2CH2S)3]−. A study of the redox behaviour of [MoO(SCH2CH2S)2]− by cyclic voltammetry using convolution analysis. Transition Met Chem 15, 177–182 (1990). https://doi.org/10.1007/BF01038371
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01038371