Summary
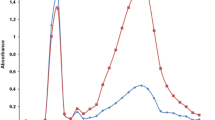
A study has been made of the possibility of using propidium iodide (PI) for the cytofluorometric determination of DNA in fixed smears. A preliminary spectrofluorometric study made it possible to define the optimal conditions for the excitation wavelength and the intensity of the emitted fluorescence. The stoichiometry and specificity of the DNA-PI bond were studied in different materials and for different fixation procedures. In order to check whether RNA might interfere, it was extracted enzymatically from test preparations selectively. The data from such treated sections were not significantly different from those obtained by means of the fluorescent Feulgen reaction (Pararosaniline-SO2) on the same material. On the other hand, some of the advantages of the PI method are important: (a) the time required for making ready and staining the preparations is very short, and in any case is considerably shorter than the feulgen method; (b) the high quantum yield of the DNA-PI complex induces very high fluorescence intensities which can, therefore, be easily measured, even with low sensitivity instruments; (c) the spectral conditions are particularly favourable for excluding the inner filter effect from the measurement; (d) the photo-decomposition is considerably lower compared to that found in preparations stained by the conventional Feulgen method. The wise possibility of excitation (from the u.v to the green), together with the limited extent of the emission band (which is mainly in the red) are also conditions that are particularly favourable for obtaining multi-parametric determinations simultaneously from the same cell.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anderson, K. J. &Skagen, D. W. (1977) Flurometric determination of DNA in fixed tissue using ethidium bromide.Analyt. Biochem. 83, 703–8.
Angerer, L. M. &Moudrianakis, E. N. (1972) Interaction of ethidium bromide with whole and selectively deproteinized deoxyribonucleoproteins from calf thymus.J. molec. Biol. 63, 505–21.
Barlogie, B., Zante, J., Drewinko, B., Schumann, J., Büchner, T., Göhde, W., Hart, J. S. &Johnston, D. A. (1976) The use of mithramycin in Pulse-Cytophotometry (PCP). InPulse Cytophotometry (edited bySchumann, J., Göhde, W. andBüchner, T.), Vol. II, pp. 125–136. Ghent: European Press.
Bittmann, R. (1969) Studies of the binding of ethidium bromide to transfer ribonucleic acid.J. molec. Biol. 46, 251–68.
Böhm, N. &Sprenger, E. (1968). Fluorescence, cytophotometry: A valuable method for the quantitative determination of nuclear Feulgen-DNA.Histochemie 16, 100–18.
Cova, S., Prenna, G. &Mazzini, G. (1974). Digital microspectrofluometry by multichannel scaling and single photon detection.Histochem. J. 6, 279–99.
Crissman, H. A., Mullaney, P. F. &Steinkamp, J. A. (1975) Methods and applications of flow systems for analysis and sorting of mammalian cells. InMethods in Cell Biology (edited byPrescott, D. M.), Vol. 9, pp. 179–246. New York: Academic Press.
Crissman, H. A. &Steinkamp, J. A. (1973). Rapid, simultaneous measurement of DNA, protein and cell volume in single cells from large mammalian cell populations.J. Cell Biol. 59, 766–71.
Crissman, H. A. &Steinkamp, J. A. (1976) Comparison of quantitative fluorescence staining methods for flow-system analysis of DNA, protein, and nuclear-to-cytoplasmic size in cultured cells, tumor cells, and human gynecological specimens. InPulse Cytophotometry (edited by,Schumann, J., Göhde, W. andBüchner, T.) Vol. II, pp. 112–124. Ghent: European Press.
Crissman, H. A. &Tobey, R. A. (1974) Cell-cycle analysis in 20 minutes.Science, N.Y. 184, 1297–8.
Dittrich, W. &Göhde, W. (1969) Impulsfluometric bei Einzellen in Suspensionen.Z. Naturforsch. 24, 221–8.
Fried, J., Perez, A. G. &Clarkson, B. D. (1976) Flow cytofluorometric analysis of cell cycle distributions using propidium iodide. Properties of the method and mathematical analysis of the data.J. Cell Biol. 71, 172–81.
Göhde, W., Spies, I., Schumann, J., Büchner, T. &Kleincdöpke, G. (1976) Two parameter analysis of DNA and protein content of tumor cells. InPulse Cytophotometry (edited bySchumann, J., Göhde, W. andBüchner, T.), Vol. II, pp. 27–32. Ghent: European Press.
Johannisson, E. &Thorell, B. (1977) Mithramycin fluorescence for quantitative determination of deoxyribonucleic acid in single cells.J. Histochem. Cytochem. 25, 122–8.
Krishan, A. (1975) Rapid flow cytofluorometric analysis of mammalian cell cycle by propidium iodide staining.J. Cell. Biol. 66, 188–93.
Lawrence, J. J. &Daune, M. (1976) Ethidium bromide as a probe of conformational heterogeneity of DNA in chromatin. The role of histone H1.Biochemistry 15, 3301–7.
Lawrence, J. J. &Louis, M. (1974) Ethidium bromide as a probe of chromatin structure.FEBS Lett. 40, 9–13.
LePecq, J. B. &Paoletti, C. (1967) A fluorescent complex between ethidium bromide and nucleic acids.J. molec. Biol. 27, 87–106.
Mansson, P. E. &Pero, R. W. (1978) Double stranded RNA in heterogenous nuclear RNA from normal and chronic lymphocytic leukemic lymphocytes.Cancer Lett. 4, 99–107.
Noeske, K. (1975) Comparison of Pulse-Cytophotometric DNA values and of nuclear DNA amounts of different classes of vertebrates. InPulse Cytophotometry (edited byHaanen, C. A. M., Hillen, H. F. P. andWessels, J. M. C.), Vol. I, pp. 36–41. Ghent: European Press.
Olmstead, J. III &Kearns, D. R. (1977) Mechanisms of ethidium bromide fluorescence enhancement on binding to nucleic acids.Biochemistry 16, 3647–54.
Patnaik, R. &Taylor, M. W. (1973) Nuclear double-stranded ribonucleic acid of mammalian cells. Characteristics and biosynthesis.Biochemistry 12, 1990–5.
Ploem, J. S. (1967) The use of vertical illuminator with interchangeable dichroic mirrors for fluorescence microscopy with incident light.Z. wiss. Mikrosk. 68, 129–42.
Prenna, G., Bottiroli, G. &Mazzini, G. (1977) Cytofluorometric quantifications of the activity and reaction kinetics of acid phosphatase.Histochem. J. 9, 15–30.
Prenna, G., Leiva, S. &Mazzini, G. (1974a) Quantitation of DNA by cytofluorometry of the conventional Feulgen reaction.Histochem. J. 6, 467–89.
Prenna, G., Mazzini, G., Bottiroli, G. &Freitas, I. (1976). Automated determination of DNA cellular content (Feulgen) improved by using BBT-SO2 in flow cytofluorometry. InPulse Cytophotometry (edited bySchumann, J., Göhde, W. andBüchner, T.), Vol. II, pp. 88–95 Ghent: European Press.
Prenna, G., Mazzini, G. &Cova, S. (1974b) Methodological and instrumentational aspects of cytofluorometry.Histochem. J. 6, 259–78.
Ruch, F. (1966) Determination of DNA content by microfluorometry. InIntroduction to Quantitative Cytochemistry (edited byWied, G. L.), pp. 281–94. New York, London: Academic Press.
Severin, E. (1975) Comparative microfluorometric investigation by MPV and ICP on liver cells and synchronized tumor cells. InPulse Cytophotometry (edited byHaanen, C. A. M., Hillen, H. F. P. andWessels, J. M. C.), pp. 42–9. Ghent: European Press.
Torelli, U., Torelli, G. &Cadossi, R. (1975) Double-stranded ribonucleic acid in human leukemic blast cells.Eur. J. Cancer 11, 117–21.
Trujillo, T. T. &Van Dilla, M. A. (1972) Adaptation of the fluorescent Feulgen reaction to cells in suspension for flow microfluorometry.Acta cytol. 16, 26–30.
Van Dilla, M. A., Trujillo, T. T., Mullaney, P. F. &Coulter, J. R. (1969) Cell microfluorometry: a method for rapid fluorescence measurement.Science 163, 1213–4.
Vindeløv, L. L. (1977) Flow microfluorometric analysis of nuclear DNA in cell from solid tumor and cell suspension. A new method for rapid isolation and staining of nuclei.Virchows Arch. B Cell Path. 24, 227–42.
Zante, J., Schumann, J., Barlogie, B., Göhde, W. &Büchner, T. (1976) New preparing and staining procedures for specific and rapid analysis of DNA distribution. InPulse Cytophotometry (edited bySchumann, J., Göhde, W. andBüchner, T.), Vol. II, pp. 97–106. Ghent: European Press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mazzini, G., Giordano, P., Montecucco, C.M. et al. A rapid cytofluorometric method for quantitative DNA determination on fixed smears. Histochem J 12, 153–168 (1980). https://doi.org/10.1007/BF01024546
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01024546