Summary
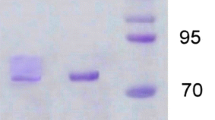
Laccase was purified from culture broth ofPleurotus ostreatus mycelium. The enzyme was a single protein of Mr 59000, pI 2.9 and was active on o-diphenyl substrates. Amino acid composition and N-terminal sequence (15 residues) were determined. Polyclonal anti-laccase antibodies were obtained.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bonner, W. M. and Laskey, R. A. (1974).Eur. J. Biochem. 46, 83–88
Bradford, M. M. (1976).Anal. Biochem. 72, 248–254.
Ishihara, T. (1980) The role of laccase in lignin biodegradation. InLignin biodegradation: microbiology, chemistry and potential applications, Kent Kirk, T., Higuchi, T. and Chang, H. eds. vol. II, pp. 17–31. CRC Press, Inc. Boca Rator, FL.
Jackson, P. and Thompson, R. J. (1984),Electrophoresis 5, 35–42
Laemmli, M. (1970).Nature 227, 680–685.
Luna, M., Poerio, E. and Badiani, M. (1983).Agrochimica 27, 44–50
Pucci, P., Sannia, G. and Marino, G. (1983).J. Chromat. 270, 371–377
Sannia, G., Abrescia, P., Colombo, M., Giardina, P. and Marino, G. (1982),Biochem. Biophys. Res. Commun. 105, 444–449.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sannia, G., Giardina, P., Luna, M. et al. Laccase fromPleurotus ostreatus . Biotechnol Lett 8, 797–800 (1986). https://doi.org/10.1007/BF01020827
Issue Date:
DOI: https://doi.org/10.1007/BF01020827