Abstract
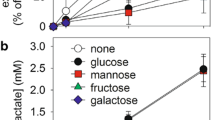
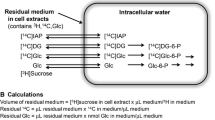
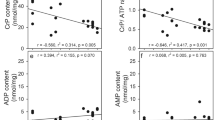
The enzymes of glycolysis and selected enzymes of the pentose phosphate pathways were measured by fluorometric methods in extracts prepared from cultures of normal cortical human astrocytes and from cultures derived from low-grade (II) or high-grade (IV) gliomas. The hexokinase and phosphofructokinase levels of the low-grade glioma-derived line were not significantly different from those of the normal astrocyte cultures. However, the activities of hexokinase and phosphofructokinase were consistently and significantly increased in the high-grade glioma-derived lines. The activity of glucose-6-phosphate dehydrogenase was significantly decreased in all glioma-derived lines and by more than 90% in the high-gradederived lines. Other enzymes of the glycolytic pathway were not significantly different from those of normal astrocytes, or they showed a variation inconsistently related to the neoplastic state. Glucose flux is not apparently regulated to a significant degree of hexokinase in glioma-derived lines, since the measured Vmax values are in substantial excess over the measured flux rates. Reversible binding of hexokinase to the particulate fraction was observed in both the normal astrocytes cultures and the high-grade glioma-derived lines. A twofold displacement of particulate hexokinase by ATP, ADP, 1-O-methylglucose, sorbito-l6-phosphate, and dibutyryl cyclic AMP was observed in the high-grade glioma-derived lines. The degree of displacement by various agents and the basal ratio of free/bound was not significantly different between the transformers and the non transformants. The hexokinase from both the gliomas and the normal astrocytes was noncompetitively inhibited by the glucose analogue 2-deoxy-rf-glucose. Phosphofructokinase activity is close to the observed glucose flux rates in both the normal astrocyte and the glioma-derived cultures. The phosphofructokinase activity of normal astrocytes is activated twofold or more by ADP, AMP, fructose-2,6-diphosphate, and P. However, these same ligands activate phosphofructokinase by less than twofold in a typical high-grade glioma-derived line. ATP, dibutyryl cyclic AMP, and citrate inhibit glioma and normal astrocytic phosphofructokinase, but the magnitude of the inhibition is much less than in the glioma-derived lines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- LCMRglc:
-
local cerebral metabolic rate for glucose
- dbcAMP:
-
dibutyryl cyclic AMP
- F2,6 diP:
-
fructose 2,6-diphosphate
- G1,6 diP:
-
glucose 1,6-diphosphate
- HK:
-
hexokinase
- PFK:
-
phosphofructokinase
- PEP:
-
phosphoenolpyruvate
- DTT:
-
dithiothreitol
- BSA:
-
bovine serum albumin
- DHAP:
-
dihydroxyacetone phosphate
- GA3P:
-
glyceraldehyde-3-phosphate
References
Bachelard, H. S. (1967). The subcellular distribution and properties of hexokinases in the guinea-pig cerebral cortex.Biochem. J. 104: 286–292.
Bigner, D. D., Bigner, S. H., Ponten, J., Westermark, B., Mahaley, M. S., Ruoslahti, E., Herschman, H., Eng, L. F., and Wikstrand, C. J. C. (1981). Heterogeneity of genotypic and phenotypic characteristics of fifteen permanent cell lines derived from human gliomas.J. Neuropathol. Exp. Neurol 40: 201–229.
Black, P. McL., Kornblith, P. L., Davison, P. F., Liszczak, T. M., Merk, L. P., Smith, B. H., McKeever, P. E., and Quindlen, E. A. (1982). Immunological, biochemical, ultrastructural and electrophysiological characteristics of a human glioblastom-derived cell culture line.J. Neurosurg. 56: 62–72
Bosca, L., Aragon, J. J., and Sols, A. (1982). Specific activation by fructose 2,6-bisphosphate and inhibition by P-enolpyruvate of ascites tumor phosphofructokinase.Biochem. Biophys. Res. Comm. 106: 486–491.
Brand, I. A., and Soling, H. D. (1975). Activation and inactivation of rat liver phosphofructokinase by phosphorylation and dephosphorylation.FEBS Lett. 57: 163–168.
Clark, M. G., and Patten, G. S. (1980). Estimation of phosphofructokinase activity in homogenates of rat heart using a new isotopic assay at very low substrate concentration.J. Mol. Cell. Card. 12: 1053–1064.
Claus, T. H., Schlumf, J. R., El-Maghrabi, M. R., Pilkis, J., and Pilkis, S. (1980). Mechanism of action of glucagon and hepatocyte phosphofructokinase.Proc. Natl. Acad. Sci. 77: 6501–6505.
Crane, R. K., and Sols, A. (1953). The association of hexokinase with particulate fractions of brain and other tissue homogenates.J. Biol. Chem. 230: 273–292.
Cummins, C. J., Smith, B. H., and Kornblith, P. L. (1984). Biochemistry of brain tumors. In Wilkins, R. H., and Rengachery, S. S. (eds.).Neurosurgery, McGraw-Hill, New York, p. 637.
DeLaPaz, R., Di Chiro, G., Smith, B. H., Kornblith, P. L., Quindlen, E. A., Sokloff, L., Brooks, R. A., Kessler, R. M., Johnston, G. S., Manning, R. G., Flynn, R. M., Wolf, A. P., Fowler, J. S., Brill, B., Blasberg, R. G., London, W. T., Sever, J. L., Kufta, C. V., Reith, K. G., Goble, J. C., and Cummins, C. J. (1981). [18F]-2-Fluoro-2-deoxyglucose positron emission tomography of human cerebral gliomas.Abstr. Am. Assoc. Neurol. Surg. 29: 30.
Di Chiro, G., DeLaPaz, R. L., Brooks, R. A., Sokoloff, L., Kornblith, P. L., Smith, B. H., Patronas, N. J., Kufta, C. V., Kessler, R. M., Johnston, G. S., Manning, R. G., and Wolf, A. P. (1982). Glucose utilization of cerebral gliomas measured by [18F]fluorodeoxyglucose and positron emission tomography.Neurology 32: 1323–1329.
Dunaway, G. A., Naqui, D., Kruep, D., Thrasher, J. R., and Morris, H. P. (1981). Phosphofructokinase isozymes of Morris hepatomas.Arch. Biochem. Biophys. 212: 1–8.
Ereci, N. M., and Silver, I. A. (1986). The role of glial cells in regulation of neurotransmitter amino acids in the external environment. I. Transmembrane electrical and ion gradients and energy parameters in cultured glial-derived cell lines.Brain Res. 26: 193–202.
Feigner, P. L., and Wilson, J. E. (1977). Effect of neutral salts on the interaction of rat brain hexokinase with the outer mitochondrial membrane.Arch. Biochem. Biophys. 182: 282–294.
Foe, L. G., and Kemp, R. G. (1982). Properties of phospho and dephospho forms of muscle phosphofructokinase.J. Biol. Chem. 257: 6368–6372.
Foe, L. G., and Trujillo, J. L. (1980). Activation of phosphofructokinase by divalent anions.Arch. Biochem. Biophys. 199: 1–6.
Galarraga, J., Loreck, D. J., Graham, J. F., DeLaPaz, R. L., Smith, B. H., Hallgren, D. H., and Cummins, C. J. (1986). Glucose metabolism in human gliomas: Correspondenceof in situ andin vitro metabolites and altered energy metabolism.Metab. Brain Dis. 1: 279–291.
Grumberger, G., Lowe, W. L., Jr., McElduff, A., and Glick, R. P. (1986). Insulin receptor of human cerebral gliomas: Structure and function.J. Clin. Invest. 77(3): 997–1005.
Kirsch, W. M. (1965). Substrates of glycolysis in intracranial tumors during complete ischemia.Cancer Res. 25: 432–439.
Kirsch, W. M., and Leitner, J. W. (1967). Glycolytic metabolites and cofactors in human cerebral cortex and white matter during complete ischemia.Brain Res. 4: 358–368.
Kornblith, P. L. (1980). Role of tissue culture in prediction of malignancy.Clin. Neurosurg. 23: 346–376.
Kornblith, P. L., and Szypko, P. (1978). Variations in response of human brain tumors to BCNUin vitro. J. Neurosurg.48: 580–586.
Long, C. (1952). Studies involving enzymic phosphorylation. I. The hexokinase activities of rat tissue.Biochem. J. 50: 407–415.
Loreck, D. J., Cummins, C. J., Galarraga, J., and Phang, J. M. (1987) Regulation of the pentose phosphate pathway in human astrocytes and gliomas.Metab. Brain Dis. 2: 31–46.
Lowry, O. H., and Passonneau, J. V. (1964). The relationships of substrates and enzymes of glycolysis in brain.J. Biol. Chem. 239: 31–42.
Lowry, O. H., and Passonneau, J. V. (1972).A Flexible System of Enzymatic Analysis, Academic Press, New York.
Lowry, O. H., Rosebrough, N., Fair, A., and Randall, R. (1951). Protein measurement using the Folin phenol reagent.J. Biol. Chem. 193: 265–275.
Lowry, O. H., Passonneau, J. V., Hasselberger, F. X., and Schulz, D. W. (1964). Effect of ischemia on known substrates and cofactors of the glycolytic pathway in brain.J. Biol. Chem. 239: 18–30.
Lowry, O. H., Berger, S. J., Carter, J. C., Chi, M. M.-Y., Manchester, J. K., Knor, J., and Pusateri, M. E. (1983). Diversity of metabolic patterns in human brain tumors: Enzymes of energy metabolism and related metabolites and cofactors.J. Neurochem. 41: 994–1010.
Naruse, S., Horikawa, Y., Tanaka, C., Higuchi, T., Ueda, S., Hirakawa, K., Nishikawa, H., and Watari, H. (1985). Observations of energy metabolism in neuroectodermal tumors usingin vivo 31P-NMR.Magnet. Reson. Imag. 3(2): 117–123.
Newsholme, E. A., Sugden, P. H., and Williams, T. (1977). Effect of citrate on the activities of 6-phosphofructokinase from nervous and muscle tissues from different animals and its relationship to the regulation of glycolysis.Biochem. J. 166: 123–129.
Perria, L., Viale, G., Ibba, F., Andreussi, L., and Viale, E. (1964). Istocitochimica dei tumori endocranici.Neuropsichiat. 20: 419–523.
Pinella, M., and Luque, J. (1981). Antagonistic effect of cAMP and cGMP on the kinetic behavior of phosphofructokinase from rat erythrocytes and reticulocytes.Ada Biol. Med. Germ. 40: 707–710.
Steel, G. G. (1980). Growth kinetics of brain tumors. In Thomas, D. G., and Graham, D. I. (eds.),Brain Tumors. Scientific Basis, Clinical Investigation and Current Therapy, Butterworth, Boston, pp. 10–20.
Timperley, W. R. (1980). Glycolysis on neuroectodermal tumors. In Thomas, D. G., and Graham, D. I. (eds.),Brain Tumors: Scientific Basis, Clinical Investigation and Current Therapy, Butterworth, Boston, pp. 145–167.
van Veelen, C. W. M., Verbiest, H., Zulch, K. J., van Ketel, B., van der Vlist, M. J. M., and Vlug, A. M. C. (1982). Pyruvate kinase in human brain tumors. Significance in the treatment of gliomas.Ada Neurochirug. 61: 145–159.
Weber, G. (1977a). Enzymology of cancer cells. I.N. Engl. J. Med. 296: 486–493.
Weber, G. (1977b). Enzymology of cancer cells. II.N. Engl. J. Med. 296: 541–550.
Wilson, J. E. (1978). Ligand-induced conformations of rat brain hexokinase: Studies with hexoses, hexose-6-phosphates, and nucleoside trisphosphates leading to development of a new model for the enzyme.Arch. Biochem. Biophys. 185: 88–99.
Wollemann, M. (1971). Biochemistry of brain tumors. In Lajtha, A. (ed.),Handbook of Neurochemistry, Vol. 7, Plenum Press, New York, p. 515.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dominguez, J.E., Graham, J.F., Cummins, C.J. et al. Enzymes of glucose metabolism in cultured human gliomas: Neoplasia is accompanied by altered hexokinase, phosphofructokinase, and glucose-6-phosphate dehydrogenase levels. Metab Brain Dis 2, 17–30 (1987). https://doi.org/10.1007/BF00999506
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00999506