Abstract
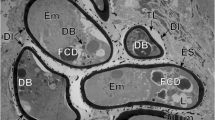
Eggshells ofMacracanthorhynchus hirudinaceus (Archiacanthocephala) were investigated for their fine structure as well as their chemical composition. The acanthor larvae are surrounded by four eggshells (E1–4) separated by interstices of low electron density (G1–4). As these envelopes are secreted in different sequences and are reinforced to different degrees, their appearance varies throughout development. The outermost eggshell (E1) of this species has a tripartite appearance; it contains neither chitin nor keratin. Keratin appears in E2 and E3. It was localized electron microscopically using anti-keratin and, for the first time by fluorescence microscopy with the bromobimane reaction. Keratin occurs in two forms: in the second envelope (E2) it consists of twisted struts of filaments, whereas in the innermost sublayer of the third envelope (E3) it shows a conspicuous cross-striation; in the first and second sublayers of E3, neither keratin nor a discernible structure is present. Chitin occurs in the innermost layer (E4). The interstices G1, G3 and G4 seem to contain glycoproteins, whereas interstice G3 seems to contain some type of carbohydrate. After the extraction of proteins including keratin with sodium dodecyl sulfate (SDS) and dithiothreitol (DTE) only layers E1 and E4 remained.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allen K, Neuberger A, Sharon N (1973) The purfication, composition and specificity of wheat germ agglutinin. Biochem J 131:155–162
Anantaraman S, Ravindranath MH (1976) Histochemical characteristics of the egg envelopes ofAcanthosentis sp. Z Parasitenkd 48:227–238
Arroyo-Begovich A, Carabez-Trejo A (1982) Location of chitin in the cyst wall ofEntamoeba invadens with colloidal gold tracers. J Parasitol 68:253–258
Brand T von (1940) Further observations upon the composition of the Acanthocephala. J Parasitol 26:301–307
Danielsohn P, Nolte A (1987) Bromobimanes—fluorescent labeling agents for histochemical detection of sulfur containing neuropeptides in semithin sections. Histochemistry 86:281–285
Edmonds SJ (1966) Hatching of the eggs ofMoniliformis dubius. Exp Parasitol 19:216–226
Fryer PR, Wells C, Ratcliff A (1983) Technical difficulties overcome in the use of Lowicryl 4KM EM embedding resin.-Histochemistry 77:141–143
Gainer H, Kosower NS (1980) Histochemical demonstration of thiels and disulfides by the fluorescent labeling agent, monobromobimane: An application to the hypothalamo-neurohypophysial system. Histochemistry 68:309–315
Geoghegan WD, Ackerman GA (1977) Adsorption of horseradish peroxidase, ovomucoid and anti-immunoglobulin to colloid gold for the indirect detection of Concanavalin A, wheat germ agglutinin and goat antihuman immunoglobulin G on cell surfaces at the electron microscopic level: a new method, theory and application. J Histochem Cytochem 25:1187–1200
Goodman SL, Hodges GM, Trejdosiewicz LK, Livingston DC (1981) Colloidal gold markers and probes for routine application in microscopy. J Microsc 123:201–213
Horisberger M (1979) Evaluation of colloidal gold as a cytochemical marker for transmission and scanning electron microscopy. Biol Cell 36:253–258
Horisberger M, Rosset J (1977) Colloidal gold, a useful marker for transmission and scanning electron microscopy. J Histochem Cytochem 25:295–305
Horisberger M, Vonlanthen M (1977) Location of mannan and chitin on thin sections of budding yeasts with gold markers. Arch Microbiol 115:1–7
Jeuniaux Ch (1963) Chitine et chitinolyse. Masson, Paris
Kosower NS, Kosower EM, Newton GL, Renney HM (1979) Bimane fluorescent labels: labeling of normal human red cells under physiological conditions. Proc Natl Acad Sci USA 76:3382–3386
Marchand B (1984) A comparative ultrastructural study of the shell surrounding the mature acanthor larvae of the 13 acanthocephalan species. J Parasitol 70:886–901
Monne L (1964) Chemie und Bildung der Embryophoren vonPolymorphus botulus Van Cleave (Acanthocephala). Z Parasitenkd 25:148–156
Peters W, Latka I (1986) Electron microscopic localization of chitin using colloidal gold labelled with wheat germ agglutinin. Histochemistry 84:155–160
Peters W, Kolb H, Kolb-Bachofen V (1983) Evidence for a sugar receptor (lectin) in the peritrophic membrane of the blowfly larva,Calliphora erythrocephala Mg. (Diptera). J Insect Physiol 29:275–280
Roth J (1983) Application of lectin-gold complexes for electron microscopic localization of glycoconjugates on thin sections. J Histochem Cytochem 31:987–999
Taraschewski H, Peters W (1991a) Comparative investigations of the morphology and chemical composition of the eggshells of Acanthocephala. II. Palaeacanthocephala. In press
Taraschewski H, Peters W (1991b) Comparative investigations of the morphology and chemical composition of the eggshells of Acanthocephala. III. Eoacanthocephala. In press
Thiéry JP (1967) Mise en évidence des polysaccharides sur coupes fines en microscopie électronique. J Microsc 6:987–1018
Tronchin G, Poulain D, Herbaut J, Biguet J (1981) Localization of chitin and the cell wall ofCandida albicans by means of wheat germ agglutinin. Fluorescence and ultrastructural studies. Eur J Cell Biol 26:121–128
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. J. Eckert (Zürich) on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Peters, W., Taraschewski, H. & Latka, I. Comparative investigations of the morphology and chemical composition of the eggshells of Acanthocephala. Parasitol Res 77, 542–549 (1991). https://doi.org/10.1007/BF00928424
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00928424