Abstract
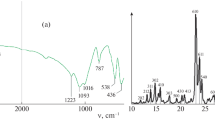
The effect of Mo loading, calcination temperature, reaction temperature and space velocity on the catalytic performance of methane dehydrogenation and aromatization without using oxidants over Mo/HZSM-5 has been studied. The XRD and BET measurements show that Mo species are highly dispersed in the channels of the HZSM-5 zeolite, resulting from the interaction between the Mo species and the zeolite, which also leads to a decrease in its crystallinity. The Brønsted acidity, the channel structure and the state and location of Mo species in the zeolite seem to be crucial factors for its catalytic performance. It was found that 2% Mo/HZSM-5 calcined at 773 K showed the best aromatization activity among the tested catalysts, the methane conversion being 9% at 1013 K with the selectivity to aromatics higher than 90%. The experimental results obtained from the variation of space velocity gave evidence that ethylene is an initial product. On the basis of these results a possible mechanism for methane dehydrogenation and aromatization has been proposed in which both the heterolytic splitting of methane in a solid acid environment and a molybdenum carbene-like complex as an intermediate are of significance.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G.A. Olah, Y. Halpern, J. Shen and Y.K. Mo, J. Am. Chem. Soc. 93 (1971) 1251.
G.A. Olah and J.A. Olah, J. Am. Chem. Soc. 93 (1971) 1256.
D.E. Webster, Adv. Organometall. Chem. 15 (1977) 147.
J.H. Lunsford,Proc. 10th Int. Congr. on Catalysis, Part A, eds. L. Guczi, F. Solymosi and P. Tétényi (Elsevier, Amsterdam, 1993) p. 69.
V.R. Choudhary, V.H. Rane and S.T. Choudhari, Catal. Lett. 6 (1990) 95.
J.A. Lapszewicz and X.-Z. Jiang, Catal. Lett. 13 (1992) 103.
V.D. Sokolovskii and E.A. Mamedov, Catal. Today 14 (1992) 331.
Y.F. Chang and H. Heinemann, Catal. Lett. 21 (1993) 215.
S.A. Shepelev and K.G. Ione, React. Kinet. Catal. Lett. 23 (1983) 323.
J.R. Anderson and P. Tsai, Appl. Catal. 19 (1985) 141.
S.S. Shepelev and K.G. Ione, J. Catal. 117 (1989) 362.
S. Han, D.J. Martenak, R.E. Palermo, J.A. Pearson and D.E. Walsh, J. Catal. 136 (1992) 578.
S. Han, D.J. Martenak, R.E. Palermo, J.A. Pearson and D.E. Walsh, J. Catal. 148 (1994) 134.
O.V. Bragin, T.V. Vasina, A.V. Preobrazhenskii and Kh.M. Minachev, Izv. Ser. Khim. No. 3 (1989) 750.
J.B. Claridge, M.L.H. Green, S.C. Tsang and A.P.E. York, Appl. Catal. A 89 (1992) 103.
L. Wang, J. Huang, L. Tao, Y. Xu, M. Xie and G. Xu, Catal. Lett. 21 (1993) 35.
R. Cid, F.J. Gil Llambias, J.L.G. Fierro, A. Lopez Agudo and J. Villasenor, J. Catal. 89 (1984) 478.
K.C. Khulbe, R.S. Mann and C.D. Ajaka, Zeolites 13 (1993) 572.
J.A. Anderson, B. Pawelec and J.L.G. Fierro, Appl. Catal. A 99 (1993) 37.
Y. Dong, S. Liu, Q. Zhang, J. Liu and K. Yang, Acta Petrolei Sinica (Petroleum Processing Section) 8 (1992) 66.
G. Buckles, G.J. Hutchings and C.D. Williams, Catal. Lett. 11 (1991) 89.
S. Kowalak and J.B. Moffat, Appl. Catal. 36 (1988) 139.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xu, Y., Liu, S., Guo, X. et al. Methane activation without using oxidants over Mo/HZSM-5 zeolite catalysts. Catal Lett 30, 135–149 (1994). https://doi.org/10.1007/BF00813680
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00813680