Summary
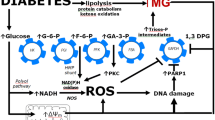
The formation of the reactiveα,β-dicarbonyl metabolite, methylglyoxal, is increased during hyperglycaemia associated with diabetes mellitus. Methylglyoxal is metabolised to S-D-lactoylglutathione and D-lactate by the glyoxalase system and to hydroxyacetone (95%) and D-lactaldehyde by aldose reductase. Methylglyoxal and hydroxyacetone bind and modify protein, producing fluorescent products. Red blood cell activities of glyoxalase enzymes are risk factors for the development of clinical complications of diabetes. Aldose reductase inhibitors decrease the concentration of methylglyoxal in experimental diabetic rats to normal levels, aminoguanidine and L-arginine scavenge methylglyoxal; these effects may be involved in their prospective preventive therapy of diabetic complications. Biochemical and clinical evidence suggests that the metabolism of methylglyoxal in diabetes mellitus is linked to the development of diabetic complications. A causal relationship may involve modification of protein by methylglyoxal and hydroxyacetone.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aleksandrovskii YA (1992) Antithrombin III, CI inhibitor, methylglyoxal, and polymorphonuclear leukocytes in the development of vascular complications of diabetes. Thromb Res 67: 179–189
Bayne S, Fewster JA (1956) The osones. Adv Carbohydr Chem 2: 43–96
Baynes JW (1991) Role of oxidative stress in the development of complications in diabetes. Diabetes 40: 405–412
Brownlee M, Vlassara H, Kooney A, Ulrich P, Cerami A (1986) Aminoguanidine prevents diabetes-induced arterial wall protein cross-linking. Science 232: 1629–1632
Brownlee M, Cerami A, Vlassara H (1988) Advanced glycation endproducts in tissue and the biochemical basis of diabetic complications. N Engl J Med 318: 1315–1321
Bunzli HF, Bosshard HR (1971) Modification of a single arginine residue in insulin with phenylglyoxal. Hoppe-Seyler's Z Physiol Chem 352: 1180–1182
Cameron NE, Cotter MA, Love A (1992) Effects of aminoguanidine on peripheral nerve function and polyol pathway metabolites in streptozotocin-diabetic rats. Diabetologia 35: 946–950
Cheung ST, Fonda ML (1979) Reaction of phenylglyoxal with arginine. Effect of buffers and pH. Biochem Biophys Res Comm 90: 940–947
Fuller JH, Kean H, Jarret RJ, Omer V, Meade TW, Chakrabart R, North WRS, Stirting Y (1979) Hemostatic variables associated with diabetes and its complications. Br Med J 2: 964–966
Gabbay KH (1973) The sorbitol pathway and the complications of diabetes. N Engl J Med 288: 831–836
Gonzalez AV, Sochor M, McLean P (1982) The effect of an aldose reductase inhibitor (sorbinil) on the level of metabolites in lens of diabetic rats. Diabetes 32: 482–485
Hallfrisch J (1990) Metabolic effects of dietary fructose. FASEB J 4: 2652–2660
Hirsch J, Petrakova E, Feather MS (1992) The reaction of dicarbonyl sugars with aminoguanidine. Carbohydr Res 232: 125–130
Horiuchi S, Murakami M, Takata K, Morimo Y (1986) Scavenger receptor for aldehydemodified proteins. J Biol Chem 261: 4962–4966
Janne J, Alhonen-Hongisto L, Nikula P, Elo H (1985) S-Adenosylmethionine decarboxylase as a target of chemotherapy. Adv Enzyme Regul 24: 125–139
Lo TWC, Selwood T, Thornalley PJ (1993) Modification of plasma protein by methylglyoxal under physiological conditions. Prevention by aminoguanidine and L-arginine. Amino Acids 5: 172 (abstract)
Lubec G, Vierhapper H, Bailey AJ, Damjanic P, Fasching P, Sims TJ, Kampel D, Popow C, Bartosch B (1991) Influence of L-arginine on glucose-mediated collagen cross link precursors in patients with diabetes mellitus. Amino Acids 1: 73–80
McCann VJ, Davis RE, Welborn TA, Constable J, Beale DJ (1981) Glyoxalase phenotypes in patients with diabetes mellitus. Aust NZ J Med 11: 380–382
McLellan AC, Phillips SA, Thornalley PJ (1992) Fluorimetric assay of D-lactate. Anal Biochem 206: 12–16
McLellan AC, Thornalley PJ, Benn J, Sonksen PH (1993) The glyoxalase system in clinical diabetes mellitus and correlation with clinical complications. Clin Sci (submitted)
Menzel EJ, Reihsner R (1991) Alterations of biochemical and biomechanical properties of rat tail tendons caused by non-enzymatic glycation and their inhibition by dibasic amino acids arginine and lysine. Diabetologia 43: 12–16
Phillips SA, Thornalley PJ (1993a) The formation of methylglyoxal from triose phosphates. Investigation using a specific assay for methylglyoxal. Eur J Biochem 212: 101–105
Phillips SA, Thornalley PJ (1993b) Formation of methylglyoxal and D-lactate in human red blood cellsin vitro. Biochem Soc Trans 21: 163S
Phillips SA, Mirrlees D, Thornalley PJ (1993) Modification of the glyoxalase system in streptozotocin-induced diabetic rats. Effect of the aldose reductase inhibitor Statil. Biochem Pharmacol 46: 805–811
Pompliano DL, Peyman A, Knowles JR (1990) Stabilization of a reaction intermediate as a catalytic device: definition of the functional role of the flexible loop in triosephosphate isomerase. Biochemistry 29: 3186–3194
Ray S, Ray M (1983) Formation of methylglyoxal from aminoacetone by amine oxidase from goat plasma. J Biol Chem 258: 3461–3462
Ray M, Ray S (1987) Aminoacetone oxidase from goat liver. J Biol Chem 262: 5974–5977
Reichard GA, Skutches CL, Hoeldtke RD, Owen OE (1986) Acetone metabolism in humans during diabetic ketoacidosis. Diabetes 35: 668–674
Richard JP (1991) Kinetic parameters for the elimination reaction catalyzed by triosephosphate isomerase and an estimation of the reaction's physiological significance. Biochemistry 30: 4581–4585
Selwood T, Thornalley PJ (1993) Binding of methylglyoxal to albumin and formation of fluorescent adducts. Inhibition by arginine,N α -acetylarginine and aminoguanidine. Biochem Soc Trans 21: 170F
Smith PR, Thornalley PJ (1992) Mechanism of the degradation of non-enzymatically glycated proteins under physiological conditions. Studies with the model fructosamine,N ε -(1-deoxy-D-fructos-l-yl)hippuryl-lysine. Eur J Biochem 210: 729–739
Smith PR, Saeed S, Selwood T, Thornalley PJ (1992) Pharmacological intervention to prevent the development of diabetic complications. Aminoguanidine diverts the degradation of lysyl-fructosamine to produce unmodified lysine and scavenges methylglyoxal. Diabetic Med 9 [Suppl] 1: 31A
Soulis-Liparota T, Cooper M, Parazoglou D, Clarke B, Jerums G (1991) Retardation by aminoguanidine of development of albuminuria, mesangial expansion, and tissue fluorescence in streptozotocin-induced diabetic rat. Diabetes 40: 1328–1334
Takahashi K (1977a) The reaction of phenylglyoxal and related reagents with amino acids. J Biochem 81: 395–402
Takahashi K (1977b) Further studies on the reaction of phenylglyoxal related reagents with proteins. J Biochem 81: 403–414
Thornalley PJ (1985) Monosaccharide autoxidation in health and disease. Environ Health Perspect 64: 297–307
Thornalley PJ (1988) Modification of the glyoxalase system in human red blood cells by glucosein vitro. Biochem J 254: 751–755
Thornalley PJ (1990) The glyoxalase system: new developments towards functional characterisation of a metabolic pathway fundamental to biological life. Biochem J 269: 1–11
Thornalley PJ, Hooper NI, Jennings PE, Florkowski CM, Jones AF, Lunec J, Barnett AH (1989) The human red blood cell glyoxalase system in diabetes mellitus. Diab Res Clin Pract 7: 115–120
Vander Jagt DL (1993) Glyoxalase II: molecular characteristics, kinetics and mechanism. Biochem Soc Trans 21: 522–527
Vander Jagt DL, Robinson B, Taylor KT, Hunsaker LA (1992) Reduction of trioses by NADPH-dependent aldo-keto reductase. J Biol Chem 267: 4364–4369
Vlassara H, Brownlee M, Manogue KR, Dinarello CA, Pasagain A (1988) Cachectin/TNF and IL-1 induced by glucose-modified proteins: role in normal tissue remodelling. Science 240: 1546–1548
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thornalley, P.J. Methylglyoxal, glyoxalases and the development of diabetic complications. Amino Acids 6, 15–23 (1994). https://doi.org/10.1007/BF00808119
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00808119