Summary
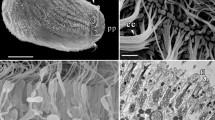
The buds ofRadiospongilla cerebellata are formed asexually. Budding can be induced experimentally by injuring the sponge. The first sign of budding is a slight elevation of some surface areas, which proceed to rise rapidly so that they soon protrude conspicuously from the surface of the sponge. As a bud develops, the broad base joining it to the mother sponge narrows to a stalk, which finally breaks. The free buds drift in the water for 15–20 min and then settle, forming new sessile sponges. The buds, 1.5–2.5 mm in diameter, have an internal organization identical with that of the mother sponge. They are enclosed in a layer of pinacoderm perforated by dermal pores. Under the pinacorderm there is a shallow subdermal space, which is in communication with the incurrent canals leading to the choanocyte chambers. The water sucked into these chambers proceeds into the excurrent canal system and emerges from the sponge through the oscular tube. Spicules projecting radially from the bud bear apical tufts of microscleres. The skeletal spicules of the buds, like their choanocyte chambers, are smaller than those in the mother sponge. The chambers expand to their mature size by choanocyte mitosis. Buds and sponges are colored green by intracellular symbiotic algae of the genusChlorella.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Boury-Esnault N (1970) Un phénomène de bourgeonnement externe chez l'épongeAxinella damicornis (Esper.) Cah Biol Mar 11:491–496
Connes R (1967) Structure et développement des bourgeons chez l'éponge siliceuseTethya lyncurium Lamarck. Arch Zool Exp Gén 108:157–195
Connes R (1968) Étude histologique, cytologique et expériementale de la régénération et de la reproduction asexuée chezTethya lyncurium Lamarck (=T. aurantium Pallas) (Demosponges). Thèse Univ Montpellier, pp 1–193
De Vos L (1965) Le bourgeonnement externe de l'épongeMycale contarenii. Bull Mus Nat Hist Natur (Paris) 37:548–555
Langenbruch PF (1984) Vergleichende rasterelektronenmikroskopische Darstellung der Gemmulaschalen vonEphydatia fluviatilis, E. muelleri undSpongilla fragilis (Porifera). Zoomorphology 104:79–85
Penney JT, Racek AA (1968) Comprehensive revision of a worldwide collection of freshwater sponges (Porifera: Spongillidae). Bull US Nat Mus 272:1–184
Saller U (1988) Oogenesis and larval development ofEphydatia fluviatilis (Porifera, Spongillidae). Zoomorphology 108:23–28
Saller U (1989) Microscopical aspects on symbiosis ofSpongilla lacustris (Porifera, Spongillidae) and green algae. Zoomorphology 108:291–296
Saller U, Weissenfels N (1985) The development ofSpongilla lacustris from the oocyte to the free larva (Porifera, Spongillidae). Zoomorphology 105:367–374
Simpson TL (1984) The Cell Biology of Sponges. Springer, Berlin Heidelberg New York
Weissenfels N (1982a) Rasterelektronenmikroskopische Histologie von spongiösem Material. Microsc Acta 85 (4):345–350
Weissenfels N (1982b) Bau und Funktion des SüßwasserschwammsEphydatia fluviatilis L. (Porifera). IX. Rasterelektronenmikroskopische Histologie und Cytologie. Zoomorphology 100:75–87
Weissenfels N (1989) Biologie und Mikroskopische Anatomie der Süßwasserschwämme (Spongillidae) G. Fischer Verlag, Stuttgart
Weissenfels N, Langenbruch P-F (1985) Langzeitkulturen von Süßwasserschwämmen (Porifera, Spongillidae) unter Laborbedingungen. Zoomorphology 105:12–15
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saller, U. Formation and construction of asexual buds of the freshwater spongeRadiospongilla cerebellata (Porifera, Spongillidae). Zoomorphology 109, 295–301 (1990). https://doi.org/10.1007/BF00803569
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00803569