Abstract
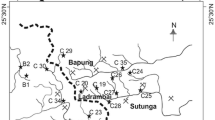
Osheepcheon Creek running through the Dogyae area is being polluted by the influx of the abandoned coal mine drainage. Generally, the more polluted water has lower pH and Eh and higher conductivity values. The concentrations of Mg, Ca, Fe, SO4, and some trace elements, such as Cd, Co, Cr, Mo, Ni, Pb, Rb, Sr, U and Zn, are tens to hundreds of times more concentrated in the abandoned coal mine drainage than in the unpolluted streamwater. However, most immobile toxic pollutants from the mine drainage are quickly removed from the streamwater by the precipitation of amorphous Fe hydroxide and sorption on the precipitated Fe hydroxide. The fast removal of the pollutants from the streamwater maintains the water quality of the creek as acceptable at most places along the stream path, except where the abandoned coal mine drainage flows in. However, the creek has the potential of deteriorating quickly if the mine drainage is allowed to be continuously combined with the streams. A function of pH, Eh, and conductivity has been developed with discriminant function analysis for the purpose of easy, fast, and inexpensive measurement of the degrees of pollution of the streams. The estimated pollution of the streams with the discriminant function are consistent with what the chemical compositions of the water samples indicate. The pollution map of the study area was constructed from the calculated scores with the discriminant function. The pollution map suggests that the pollutants mainly come from the west side of Osheepcheon Creek. Thus, the abandoned coal mine drainage from the west side has to be appropriately treated as soon as possible to prevent Osheepcheon Creek from being further polluted. Considering the topography, climate, and the amount of the mine drainage, an active treatment method is recommended.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ahn JH, Yun JM, Lee HG, and Kim DH (1990) Study on the counterplan to the increased water drainage from the abandoned mines. Korea Mining Promotion Corporation, Seoul, Report 90-1. 321 pp
Allison JD, Broen DS, and Novo-Gradac KJ (1991) MINTEQA2/PRODEFA2, A geochemical assessment model for environmental systems: Version 3.0 user's manual. EPA/600/3-91/021. 105 pp
Anderson PR and Benjamin MM (1985) Effects of silicon on the crystallization and adsorption properties of ferric oxides. Environ Sci Technol 19:1048–1053
APHA, AWWA, and WEF (1992) Standard methods for the examination of water and waste water, 18th ed. Washington, DC: APHA Pub.
Cheong CH (1969) Stratigraphy and paleontology of the Samcheok coal field, Kangwon-Do, Korea (I). J Geol Soc Korea 5:13–56
Davis JC (1986) Statistics and data analysis in geology, 2nd ed. New York: John Wiley & Sons. 646 pp
Lee JH, Lee SH, Won JK, Noh JH, Lee MW, Park SI, Woo KS, Kim YH, and Lee KC (1992) Geotectonical and petrographie studies on the sedimentary, igneous, and metamorphic rocks of Taebag Area, Kangwon-Do. Korean Science and Engineering Foundation, Taejeon, Report 88-1113-01. 402 pp
Livingstone DA (1963) Chemical compositions of rivers and lakes. USGS Prof. Paper 440-G
Nordstrom DK (1982) The effect of sulfate on aluminum concentrations in natural waters: Some stability relations in the system A12O3-SO3-H2O at 298K. Geochim Cosmochim Acta 46:681–692
Nordstrom DK, Jenne EA, and Ball JW (1979). Redox equilibria of iron in acid mine waters. In: Jenne EA (Ed), Chemical modeling in aqueous systems: Speciation, sorption, solubility and kinetics. Washington, DC: American Chemical Society Symposium Series 93. pp 51–94
Powell JD (1988) Origin and influence of coal mine drainage on streams in United States. Environ Geol Water Sci 11:141–152
Sullivan PJ, Yelton JL, and Reddy KJ (1988a) Solubility relationships of aluminum and iron minerals associated with acid mine drainage. Environ Geol Water Sci 11:283–287
Sullivan PJ, Yelton JL, and Reddy KJ (1988b) Iron sulfide oxidation and the chemistry of acid generation. Environ Geol Water Sci 11:289–295
Theobald, PK Jr, Larkin HW, and Hawkins DB (1963) The precipitation of aluminum, iron and manganese at the junction of Deer Creek with Snake River in Summit County, Colorado. Geochim Cosmochim Acta 27:121–132
Uma KO (1992) Origin of acid mine drainage in Enugu. Environ Geol Water Sci 20:181–194
Webb JA and Sasowsky ID (1994) The interaction of acid mine drainage with a carbonate terrane; evidence from the Obey River, north-central Tennessee. J Hydrol 161:327–346
Wetzel RG and Likens GE (1991) Limnological analysis, 2nd ed. New York: Springer-Verlag. 391 pp
Wiggering H (1993) Sulfide oxidation-an environmental problem within colliery spoil dumps. Environ Geol 22:99–105
Wood WW (1976) Guidlines for the collection and field analysis of ground water samples for selected unstable constituents. USGS WRI Book 1, Chapter D2. pp 1–24
Zaihua L, Daoxian Y, and Zhaoli S (1991) Effects of coal mine waters of variable pH on springwater quality: A case study. Environ Geol Water Sci 17:219–225
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yu, J.Y. Pollution of Osheepcheon Creek by abandoned coal mine drainage in Dogyae area, eastern part of Samcheok coal field, Kangwon-Do, Korea. Geo 27, 286–299 (1996). https://doi.org/10.1007/BF00766698
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00766698