Summary
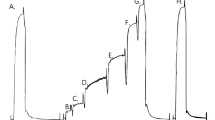
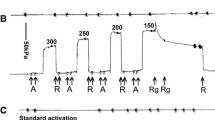
Complex stiffness data were studied over an extended range of MgATP concentrations (3 µm-5mm) in single fibres of Ca2+-activated, chemically skinned adult rabbit psoas. The data were analysed in terms of a model involving three exponential processes, the presence of which was previously observed in fully activated muscles. As fibres were transferred from a rigor solution into solutions of gradually increasing MgATP concentration, the three processes appeared sequentially, each with a uniqueK m. The order of appearance as MgATP increases is (1) the slowest of the three processes [designated process (A)], (2) the fastest of the three processes [designated (C)], and (3) process (B), which occupies the middle range of frequencies; theK ms are approximately 10 µm, 0.2mm, and 0.8mm, respectively. The single phase advance [process (A)] remaining at very low substrate concentrations was found to be better described by a distribution of rate constants than by a single rate constant. The influence of substrate concentration on these processes is examined and two parallel hydrolysis routes are suggested as a possible mechanism.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
ABBOTT, R. H. (1973) The effects of fibre length and calcium ion concentration on the dynamic response of glycerol extracted insect fibrillar muscle.J. Physiol. 231, 195–208.
BÖTTCHER, C. J. P. (1952)Theory of Electrical Polarization, pp. 363–374. Amsterdam: Elsevier.
BRANDT, P. W., CHAPPELL, E. & JEWELL, B. R. (1976) A robust transducer suitable for measuring forces of 1 µN.J. Physiol. 258, 43–4P (abstract).
BRANDT, P. W., COX, R. N. & KAWAI, M. (1980) Can the binding of Ca2+ to two regulatory sites on troponin-C determine the steep pCa/tension relation of skeletal muscle?Proc. natn. Acad. Sci. U.S.A. 77, 4717–20.
BREMEL, R. D. & WEBER, A. (1972) Cooperation within actin filament in vertebrate skeletal muscle.Nature New Biol. 238, 97–101.
CHAPLAIN, R. A. & FROMMELT, B. (1968) On the contractile mechanism of insect fibrillar flight muscle. I. The dynamics and energetics of the linearized system.Kybernetick 5, 1–17.
COLE, K. S. & COLE, R. H. (1941) Dispersion and absorption in dielectrics. I. Alternating current characteristics.J. Chem. Phys. 9, 341–51.
EASTWOOD, A. B., WOOD, D. S., BOCK, K. L. & SORENSON, M. M. (1979) Chemically skinned mammalian skeletal muscle. I. The structure of skinned rabbit psoas.Tiss. Cell 11, 553–66.
EISENBERG, E. & GREENE, L. E. (1980) The relation of muscle biochemistry to muscle physiology.Ann. Rev. Physiol. 42, 293–309.
HUXLEY, A. F. (1957) Muscle structure and theories of contraction.Prog. Biophys. 7, 255–318.
HUXLEY, A. F. (1974) Muscular contraction.J. Physiol. 243, 1–43.
HUXLEY, A. F. & SIMMONS, R. M. (1971) Proposed mechanism of force generation in striated muscle.Nature 233, 533–8.
JULIAN, F. J., SOLLINS, K. R. & SOLLINS, M. R. (1974) A model for the transient and steady-state mechanical behavior of contracting muscle.Biophys. J. 14, 546–62.
KAWAI, M. (1978) Head rotation or dissociation? A study of exponential rate processes in chemically skinned rabbit muscle fibers when MgATP concentration is changed.Biophys. J. 22, 97–103.
KAWAI, M. (1979) Effect of MgATP on cross-bridge kinetics in chemically skinned rabbit psoas fibers as measured by sinusoidal analysis technique. InCross-bridge Mechanism in Muscle Contraction (edited by SUGI, H. and POLLACK, G. H.), pp. 149–169. Tokyo: University of Tokyo Press.
KAWAI, M. & BRANDT, P. W. (1976) Two rigor states in skinned crayfish single muscle fibers.J. gen. Physiol. 68, 267–80.
KAWAI, M. & BRANDT, P. W. (1977) Effect of MgATP on stiffness measured at two frequencies in Ca2+-activated fibres.Proc. natn. Acad. Sci. 74, 4073–5.
KAWAI, M. & BRANDT, P. W. (1979) Effect of ATP buffer concentration on the mechanical rate constants in chemically skinned rabbit psoas fibers.Biophys. J. 25, 270a (abstract).
KAWAI, M. & BRANDT, P. W. (1980) Sinusoidal analysis; a high resolution method for correlating biochemical reactions with physiological processes in activated skeletal muscles of rabbit, frog and crayfish.J. Mus. Res. Cell Motil. 1, 279–303.
KAWAI, M., BRANDT, P. W. & ORENTLICHER, M. (1977) Dependence of energy transduction in intact skeletal muscles on the time in tension.Biophys. J. 18, 161–72.
KAWAI, M., COX, R. N. & BRANDT, P. W. (1981) Effect of Ca ion concentration on crossbridge kinetics in rabbit psoas fibres: Evidence for the presence of two Ca-activated states of thin filament.Biophys. J. (in press).
LYMN, R. W. & TAYLOR, E. W. (1971) The mechanism of adenosine triphosphate hydrolysis by actomyosin.Biochemistry 10, 4617–24.
MARTELL, A. E. (1964)Stability Constants of Metal-ion Complexes. London: The Chemical Society.
PODOLSKY, R. J. & NOLAN, A. C. (1973) Muscle contraction transients, cross-bridge kinetics, and the Fenn effect.Cold Spring Harb. Symp. quant. Biol. 37, 661–8.
PRINGLE, J. W. S. (1967) The contractile mechanism of insect fibrillar muscle.Prog. biophys. molec. Biol. 17, 1–60.
REUBEN, J. P., BRANDT, P. W., BERMAN, M. & GRUNDFEST, H. (1971) Regulation of tension in the skinned crayfish muscle fiber. I. Contraction and relaxation in the absence of Ca (pCa>9).J. gen. Physiol. 57, 385–407.
STEIGER, G. J., BRADY, A. J. & TAN, S. T. (1978) Intrinsic regulatory properties of contractility in the myocardium.Circulation Res. 42, 339–50.
STEIN, L. A., SCHWARZ, R. P., CHOCK, P. B. & EISENBERG, E. (1979) Mechanism of actomyosin adenosine triphosphatase. Evidence that adenosine 5′-triphosphate hydrolysis can occur without dissociation of the actomyosin complex.Biochemistry 18, 3895–909.
TONOMURA, Y. (1978) Functional implications of the two-headed structure of myosin.Sixth Int. Biophys. Cong. Abst. 74.
WHITE, D. C. S. & THORSON, J. (1972) Phosphate starvation and the nonlinear dynamics of insect fibrillar flight muscle.J. gen. Physiol. 60, 307–36.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cox, R.N., Kawai, M. Alternate energy transduction routes in chemically skinned rabbit psoas muscle fibres: a further study of the effect of MgATP over a wide concentration range. J Muscle Res Cell Motil 2, 203–214 (1981). https://doi.org/10.1007/BF00711870
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00711870