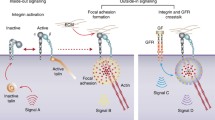
Summary
Apoptosis is a regulated process of cell death by which cells actively participate in their own destruction. In multicellular organisms, the balance between cell proliferation and apoptosis provides homeostatic control, and a regulatory failure of either event can contribute to oncogenesis. The extracellular matrix (ECM) is known to play a regulatory role in cellular growth and differentiation, but only more recently has it been recognized as a regulator of apoptosis. In these processes the major transmitters of ECM-derived signals to the cell are members of the integrin family, although the mechanical process of cell spreading also plays a role. Bothin vivo andin vitro the loss of adhesion to specific components of the ECM can lead to cell death, and such apoptosis can be induced experimentally by blocking integrin binding. Heterotypic and homotypic cell-cell adhesion can also protect from adhesion-dependent apoptosis and there is evidence to suggest that this too is integrin mediated. In addition, some integrin mediated signaling appears to promote apoptosis. The downstream mechanisms of integrin signaling causing cell death have not been greatly explored, but there is evidence from two different systems that the induction of ICE transcription and nuclear translocation of p53 are candidate processes. Alterations in integrin expression or signaling therefore are likely to contribute to tumor development by enabling escape from apoptosis. Also, the recognition of the importance of cell-cell adhesion in tumor cell survival offers the potential of developing improved drug regimes for the treatment of malignancy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ruoslahti E: Integrins. J Clin Invest 87: 1–5, 1991
Hynes RO: Integrins: versatility, modulation, and signaling in cell adhesion. Cell 69: 11–25, 1992
Bodary SC, Lipari T, Muir C, Napier M, Pitti R, McLean JW: Deletion of the cytoplasmic and transmembrane domains of GPIIbIIIa results in a functional receptor. J Cell Biol 115: 289a, 1991
O'Toole TE, Mandelman J, Forsyth J, Shattil SJ, Plow EF, Ginsberg MH: Modulation of the affinity of integrin αIIbβ3 (GPIIbIIIa) by the cytoplasmic domain of αIIb. Science 254: 845–847, 1991
Crowe DT, Chiu H, Fong S, Weissman IL: Regulation of the avidity of integrin α4β7 by the β7 cytoplasmic domain. J Biol Chem 269: 14411–14418, 1994
Elices MJ, Urry LA, Hemler ME: Receptor functions for the integrin VLA-3: fibronectin, collagen, and laminin binding are differentially influenced by ARG-GLY-ASP peptide and by divalent cations. J Cell Biol 112: 169–181, 1991
Bates RC, Rankin LM, Lucas CM, Scott JL, Krissansen GW, Burns GF: Individual embryonic fibroblasts express multiple β chains in association with the αv integrin subunit. Loss of β3 expression with cell confluence. J Biol Chem 266: 18593–18599, 1991
Dhawan J, Lichtler AC, Rose DW, Farmer SR: Cell adhesion regulates pro-alpha 1 (I) collagen mRNA stability and transcription in mouse fibroblasts. J Biol Chem 266: 8470–8475, 1991
Dike LE, Farmer SR: Cell adhesion induces expression of growth associated genes in suspension arrested fibroblasts. Proc Natl Acad Sci USA 85: 6792–6796, 1988
Ingber DE: Fibronectin controls capillary endothelial cell growth by modulating cell shape. Proc Natl Acad Sci USA 87: 3579–3583, 1990
Stoker M, O'Neill C, Berryman S, Waxman V: Anchorage and growth regulation in normal and virus transformed cells. Int J Cancer 3: 683–693, 1968
Meredith J, Fazeli B, Schwartz M: The extracellular matrix as a cell survival factor. Mol Biol Cell 4: 953–961, 1993
Frisch SM, Francis H: Disruption of epithelial cell-matrix interactions induces apoptosis. J Cell Biol 124: 619–626, 1994
Re F, Zanetti A, Sironi M, Polentarutti N, Lanfrancone L, Dejana E, Colotta F: Inhibition of anchorage-dependent cell spreading triggers apoptosis in cultured human endothelial cells. J Cell Biol 127: 537–546, 1994
Adams J, Watts F: Regulation of development and differentiation by the extracellular matrix. Development 117: 1183–1198, 1993
Kerr JFK, Wyllie AH, Currie AH: Apoptosis, a basic biological phenomenom with wider implications in tissue kinetics. Br J Cancer 26: 239–245, 1972
Cohen JJ: Apoptosis. Immunology Today 14: 126–130, 1993
Martin SJ, Green DR, Cotter TG: Dicing with death: dissecting the components of the apoptosis machinery. TIBS 19: 26–30, 1994
Schwartz LM, Osborne BA: Programmed cell death, apoptosis and killer genes. Immunology Today 14: 582–590, 1993
Cotter TG, Lennon SV, Glynn JG, Martin SJ: Cell death via apoptosis and its relationship to growth, development and differentiation of both tumour and normal cells. Anticancer Res 10: 153–1160, 1990
Williams GT: Programmed cell death: apoptosis and oncogenesis. Cell 65: 1097–1098, 1991
Vaux D, Cory S, Adams J: Bcl-2 gene promotes haemopoietic cell survival and cooperates with c-myc to immortalize pre-B cells. Nature 335: 440–442, 1988
Vaux DL, Weissman IL, Kim SK: Prevention of programmed cell death in Caenorhabditis elegans by human bcl-2. Science 258: 1955–1957, 1992
Talhouk RS, Chin JR, Unemori EN, Werb Z, Bissell MJ: Proteinases of the mammary gland: developmental regulationin vivo and vectorial secretion in culture. Development 112: 439–449, 1991
Boudreau N, Sympson CJ, Werb Z, Bissell MJ: Suppression of ICE and apoptosis in mammary epithelial cells by extracellular matrix. Science 267: 891–893, 1995
Boudreau N, Myers C, Bissell MJ: From laminin to lamin: regulation of tissue-specific gene expression by the ECM. Trends Cell Biol 5: 1–4, 1995
Kalcheim C, Barde Y-A, Thoenen H, Le Douarin N:In vivo effect of brain-derived neurotrophic factor on the survival of developing dorsal root ganglion cells. EMBO J 6: 2871–2873, 1987
Roskelley CD, Desprez PY, Bissell MJ: Extracellular matrix-dependent tissue specific gene expression in mammary epithelial cells requires both physical and biochemical signal transduction. Proc Natl Acad Sci USA 91:12378–12382, 1994
Albelda SM, Mette SA, Elder DE, Stewart R, Damjanovich L, Herlyn M, Buck CA: Integrin distribution in malignant melanoma: association of the β3 subunit with tumor progression. Cancer Res 50: 6757–6764, 1990
Seftor REB, Seftor EA, Gehlsen KR, Stetler-Stevenson WG, Brown PD, Rouslahti E, Hendrix MJC: Role of αvβ3 integrin in human melanoma cell invasion. Proc Natl Acad Sci USA 89: 1557–1561, 1992
Montgomery AMP, Reisfeld RA, Cheresh DA: Integrin αvβ3 rescues melanoma cells from apoptosis in three-dimensional dermal collagen. Proc Natl Acad Sci USA 91: 8856–8860, 1994
Raff MC: Social control on cell survival and cell death. Nature 356: 397–400, 1992
Manabe A, Coustan-Smith E, Behm FG, Raimondi SC, Campana D: Bone marrow-derived stromal cells prevent apoptotic cell death in B-lineage acute lymphoblastic leukemia. Blood 79: 2370–2377, 1992
Falk MH, Hultner L, Milner A, Gregory CD, Bornkamm GW: Irradiated fibroblasts protect Burkitt lymphoma cells from apoptosis by a mechanism independent of bcl-2. Int J Cancer 55: 485–491, 1993
Fujita N, Kataoka S, Naito M, Heike Y, Boku N, Nakajima M, Tsuruo T: Suppression of T-lymphoma cell apoptosis by monoclonal antibodies raised against cell surface adhesion molecules. Cancer Res 53: 5022–5027, 1993
Bates RC, Buret A, van Helden D, Horton MA, Burns GF: Apoptosis induced by inhibition of intercellular contact. J Cell Biol 125: 403–415, 1994
Whitehead RH, Jones JK, Gabriel A, Lukies RE: A new colon carcinoma cell line (LIM 1863) that grows as organoids with spontaneous differentiation into crypt-like structuresin vitro. Cancer Res 47: 2683–2689, 1987
Hayward IP, Whitehead RH: Patterns of growth and differentiation in the colon carcinoma cell line LIM 1863. Int J Cancer 50: 752–759, 1992
Ruoslahti E, Reed JC: Anchorage dependence, integrins and apoptosis. Cell 77: 477–478, 1994
Schmidt J, Piepenhagen P, Nelson WJ: Modulation of epithelial morphogenesis and cell fate by cell-to-cell signals and regulated cell adhesion. Semin Cell Biol 4: 161–173, 1993
Ojakian G: Tumor promoter-induced changes in the permeability of epithelial cell tight junctions. Cell 23: 95–103, 1981
Behrens J, Weidner K, Frixen U, Schipper J, Sachs M, Arakaki N, Daikuhara Y, Birchmeier W: The role of E-cadherin and scatter factor in tumor invasion and cell motility. In: Goldburg I (ed) Cell Motility. Birkhauser Verlag, Basel, 1991
Jewell K, Kapron-Bas C, Jeevaratnam P, Dedhar S: Stimulation of tyrosine phosphorylation of distinct proteins in response to antibody-mediated ligation and clustering of α3 and β6 integrins. J Cell Sci 108: 1995 [in press]
Kapron-Bras C, Fitz-Gibbon L, Jeevaratnam P, Wilkins J, Dedhar S: Stimulation of tyrosine phosphorylation and accumulation of GTP-bound p21ras upon antibody-mediated α2β1 integrin activation in T-lymphoblastic cells. J Biol Chem 268: 20701–20704, 1993
Burridge K, Turner CE, Romer LH: Tyrosine phosphorylation of paxillin and pp125FAK accompanies cell adhesion to extracellular matrix: a role in cytoskeletal assembly. J Cell Biol 119: 893–903, 1992
Schaller MD, Borgman CA, Cobb BS, Vines RR, Reynolds SB, Parsons JT: pp125FAK, a structurally distinctive proteintyrosine kinase associated with focal adhesion. Proc Natl Acad Sci USA 89: 5192–5196, 1992
Hanks SK, Calalb MB, Harper MC, Patel SK: Focal adhesion protein-tyrosine kinase phosphorylated in response to cell attachment to fibronectin. Proc Natl Acad Sci USA 89: 8487–8491, 1992
Juliano RL, Haskill S: Signal transduction from the extracellular matrix. J Cell Biol 120: 577–585, 1993
Chen Q, Kinch MS, Lin TH, Burridge K, Juliano RL: Integrin-mediated cell adhesion activates mitogen-activated protein kinases. J Biol Chem 269: 26602–26605, 1994
Morino N, Mimura T, Hamasaki K, Tobe K, Ueki K, Kikuchi K, Takehara K, Kadowaki T, Yazaki Y, Nojima Y: Matrixintegrin interaction activates the mitogen-activated protein kinase, p44erk-1 and p42erk-2. J Biol Chem 270: 269–273, 1995
Ahn NG, Seger R, Bratlien RL, Diltz CD, Tonks NK, Krebs EG: Multiple components in an Epidermal growth factorstimulated protein kinase cascade. J Biol Chem 266: 4220–4227, 1991
Lenormand P, Sardet C, Pages G, L'Allemain G, Brunet A, Pouyssegeur J: Growth factors induce nuclear translocation of MAP kinases (p42mapk and p44mapk) but not of their activator MAP kinase (p45mapk) in fibroblasts. J Cell Biol 122: 1079–1088, 1993
Ojcius DM, Zychlinsky A, Zheng LM, Young JDE: Ionophore-induced apoptosis: role of DNA fragmentation and calcium fluxes. Exp Cell Res 197: 43–49, 1991
Baffy G, Miyashita T, Williamson J, Reed JC: Apoptosis induced by withdrawal of interleukin-3 (IL-3) from an IL-3-dependent hematopoietic cell line is associated with repartitioning of intracellular calcium and is blocked by enforced bcl-2 oncoprotein production. J Biol Chem 268: 6511–6519, 1993
Uckun FM, Tuel-Ahlgren L, Song CW, Waddick K, Myers DE, Kirihara J, Ledbetter JA, Schieven GL: Ionizing radiation stimulates unidentified tyrosine-specific protein kinases in human B-lymphocyte precursors, triggering apoptosis and clonogenic cell death. Proc Natl Acad Sci USA 89: 9005–9009, 1992
Song Q, Baxter GD, Kovacs EM, Findik D, Lavin MF: Inhibition of apoptosis in human tumor cells by okadaic acid. J Cell Physiol 153: 550–556, 1992
Williams GT, Smith CA: Molecular regulation of apoptosis: genetic controls on cell death. Cell 74: 777–779, 1993
Reed JC: Bcl-2 and the regulation of programmed cell death. J Cell Biol 124: 1–6, 1994
Vaux DL, Haecker G, Strasser A: An evolutionary perspective on apoptosis. Cell 76: 777–779, 1994
Haas-Kogan DA, Kogan SC, Levu D, Dazin P, T'Ang A, Fung Y-K, Israel MA: Inhibition of apoptosis by the retinoblastoma gene product. EMBO J 14: 461–472, 1995
Smeyne RJ, Vendrell M, Hayward M, Baker SJ, Miao GG, Schilling K, Robertson LM, Curran T, Morgan JI: Continuous c-fos expression precedes programmed cell deathin vivo. Nature 363: 166–169, 1993
Wyllie AH: Death gets a brake. Nature 369: 272–273, 1994
Yuan J, Shahan S, Ledoux S, Ellis HM, Horvitz HR: The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1 beta-converting enzyme. Cell 75: 641–652, 1993
Miura M, Zhu H, Rotello R, Hartwieg EA, Juan J: Induction of apoptosis in fibroblasts by IL-1 beta-converting enzyme, a mammalian homolog of theC. elegans cell death gene ced-3. Cell 75: 653–660, 1993
Peitsch MC, Polzar B, Stephan H, Crompton T, MacDonald HR, Mannherz HG, Tschopp J: Characterization of the endogenous deoxyribonuclease involved in nucleic DNA degradation during apoptosis (programmed cell death). EMBO J 12: 371–377, 1993
Vogelstein B, Kinzler KW: p53 function and dysfunction. Cell 70: 523–526, 1992
Yonish-Rouach E, Resnitzky D, Lotem J, Sachs L, Kimchi A, Oren M: Wild-type p53 induces apoptosis of myeloid leukaemic cells that is inhibited by interleukin-6. Nature 352: 345–347, 1991
Yonish-Rouach E, Grunwald D, Wilder S, Kimchi A, May C, Lawrence JJ, May P, Oren M: p53 mediated cell death: relationship to cell cycle control. Mol Cell Biol 13: 1415–1423, 1993
Shaw P, Bovey R, Tardy S, Sahli R, Sordat B, Costa J: Induction of apoptosis by wild-type p53 in a human colon-derived cell line. Proc Natl Acad Sci USA 89: 4495–4499, 1992
Liu Z-G, Smith SW, McLaughlin KA, Schwartz LM, Osborne BA: Apoptotic signals delivered through the T-cell receptor of a T-cell hybrid require the immediate-early gene nur77. Nature 367: 281–284, 1994
Dedhar S, Rennie PS, Shago M, Leung-Hagesteijn C, Yang H, Filmus J, Hawley RG, Bruchovsky N, Cheng H, Matusik RJ, Giguere V: Inhibition of nuclear hormone receptor activity by calreticulin. Nature 367: 480–483, 1994
Dedhar S: Novel functions for calreticulin: interaction with integrins and modulation of gene expression? TIBS 19: 269–271, 1994
Leung-Hagesteijn CY, Milankov K, Michalak M, Wilkins J, Dedhar S: Cell attachment to extracellular matrix substrates is inhibited upon downregulation of expression of calreticulin, an intracellular integrin α-subunit binding protein. J Cell Sci 107: 589–600, 1994
Pignatelli M, Bodmer WF: Integrin cell adhesion molecules and colorectal cancer. J Path 162: 95–97, 1990
Klein CE, Dressel D, Steinmayer T, Mauch C, Eckes B, Kreig T, Bankert RB, Weber L: Integrin α2β1 is upregulated in fibroblasts and highly aggressive melanoma cells in three dimensional collagen lattices and mediates the reorganization of collagen I fibrils. J Cell Biol 115: 1427–1436, 1991
Natali PG, Nicotra MR, Botti C, Mottolese, Bigotti A, Segatto O: Changes in expression of α6/β4 integrin heterodimer in primary and metastatic breast cancer. Br J Cancer 66: 318–322, 1992
Raz A: Adhesive properties of metastasizing tumor cells. Ciba Found Symp 141: 323–330, 1988
Liotta LA, Kleinerman J, Saidel GM: Quantitative relationships of intravascular tumor cells, tumor vessels, and pulmonary metastases following tumor implantation. Cancer Res 34: 997–1004, 1974
Liotta LA, Kleinerman J, Saidel GM: The significance of hematogenous tumor cell clumps in the metastatic process. Cancer Res 36: 889–894, 1976
Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML, Wyllie AH: Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362: 849–852, 1993
Lowe SW, Schmitt EM, Smith SW, Osborne BA, Jacks T: p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362: 847–849, 1993
Moll UM, Riou G, Levine AJ: Two distinct mechanisms alter p53 in breast cancer: mutation and nuclear exclusion. Proc Natl Acad Sci USA 89: 7262–7266, 1992
Graham CH, Kobayashi H, Stankiewicz KS, Man S, Kapitain SJ, Kerbel RS: Rapid aquisition of multicellular drug resistance after a single transient exposure of mammary tumor cells to alkylating agents. J Natl Canc Inst 86: 975–982, 1994
Kobayashi H, Man S, Graham CH, Kapitain SJ, Teicher BA, Kerbel RS: Acquired multicellular-mediated resistance to alkylating agents in cancer. Proc Natl Acad Sci USA 90: 3294–3298, 1993
Kerbel RS, St. Croix B, Rak J, Graham C: Is there a role for ‘anti-adhesives’ as chemosensitizers in the treatment of solid tumors by chemotherapy? Bulletin de l'Institut Pasteur 1994 [in press]
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bates, R.C., Lincz, L.F. & Burns, G.F. Involvement of integrins in cell survival. Cancer Metast Rev 14, 191–203 (1995). https://doi.org/10.1007/BF00690291
Issue Date:
DOI: https://doi.org/10.1007/BF00690291