Summary
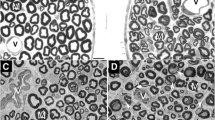
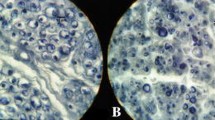
One year after beginning of the experiment seven streptozotocin-injected Wistar rats and seven controls were fixed by whole-body perfusion, the nervus radialis was dissected and processed for light and electron microscopy. After light-microscopic study standard photographs of nerve cross sections were measured by means of a semiautomatic image analyzer. The following measurements were obtained: (1) surface of fibers, axons, and myelin sheaths; (2) ratio of myelin to axon surface; and (3) percent of endoneural space. Group means and standard errors were calculated, and cumulated class distributions were made. Ultrathin sections from all animals considered morphometrically were studied qualitatively for ultrastructural changes. The quantitative study revealed in the diabetics reduction of average myelin surface, increase of endoneural space, and reduction of myelin/axon ratio. The main ultrastructural findings were lesions of Schwann and mesenchymal cells, followed by less frequent and less severe changes in axons and endothelium. These results suggest a primary Schwann cell lesion was responsible for the observed myelin reduction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bestetti G, Rossi GL (1980) Hypothalamic lesions in rats with long term streptozotocin-induced diabetes mellitus. Acta Neuropathol (Berl) 52:119–127
Bischoff A (1977) Die diabetische Neuropathie. In: Oberdisse K (Hrsg) Handbuch der inneren Medizin, Bd 7, Teil 2. Springer, Berlin Heidelberg New York, S 415–464
Clements RS Jr (1979) Diabetic neuropathy. New concepts of its etiology. Diabetes 28:604–611
Jakobsen J (1976a) Axonal dwindling in early experimental diabetes. I. A study of cross-sectioned nerves. Diabetologia 12:539–546
Jakobsen J (1976b) Axonal dwindling in early experimental diabetes. II. A study of isolated nerve fibers. Diabetologia 12:547–553
Jakobsen J (1978) Peripheral nerves in early experimental diabetes. Expansion of the endoneurial space as a cause of increased water content. Diabetologia 14:113–119
Junod A, Lambert AE, Orci L, Pictet R, Gonet AE, Renold AE (1967) Studies of the diabetogenic action of streptozotocin. Proc Soc Exp Biol Med 126:201–205
Junod A, Lambert AE, Stauffacher W, Renold AE (1969) Diabetogenic action of streptozotocin: relationship of dose to metabolic response. J Clin Invest 48:2129–2139
Powell H, Knox D, Lee S, Charters AC, Orloff M, Garrett R, Lampert P (1977) Alloxan diabetic neuropathy: electronmicroscopic studies. Neurology (Minneap) 27:60–66
Rossi GL (1975) Simple apparatus for perfusion fixation for electron microscopy. Experientia (Basel) 31:998
Sharma AK, Thomas PK (1974) Peripheral nerve structure and function in experimental diabetes. J Neurol Sci 23:1–15
Sharma AK, Thomas PK, De Molina AF (1977) Peripheral nerve fiber size in experimental diabetes. Diabetes 26:689–692
Sima AAF, Robertson DM (1979) Peripheral neuropathy in the diabetic mutant mouse. An ultrastructural study. Lab Invest 40:627–632
Spencer PS, Thomas PK (1974) Ultrastructural studies of the dying-back process. II. The sequestration and removal by Schwann cells and oligodendrocytes of organelles from normal and diseased axons. J Neurocytol 3:763–783
Yagihashi S, Nishihira M, Baba M (1979) Morphometrical analysis of the peripheral nerve lesions in experimental diabetes rats. Tohoku J Exp Med 129:139–149
Author information
Authors and Affiliations
Additional information
Supproted by the Schweizer Nationalfonds grants nos. 3.198-0.77 and 3.552-0.79
Rights and permissions
About this article
Cite this article
Zemp, C., Bestetti, G. & Rossi, G.L. Morphological and morphometric study of peripheral nerves from rats with streptozotocin-induced diabetes mellitus. Acta Neuropathol 53, 99–106 (1981). https://doi.org/10.1007/BF00689989
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00689989