Abstract
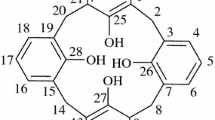
From the reaction ofp-t-butylcalix[4]arene with α-chloro-N,N-diethyl acetamide a new lipophilic ether-amide ligand (2) has been obtained in high yield. Solution studies show (2) to be a very strong cation receptor for alkali cations, especially sodium and potassium. The X-ray crystal structure determination of the free ligand (2) and two potassium complexes (KI and KSCN) shows the calix[4]arene in a fixed ‘cone’ structure and the cation completely encapsulated in a polar cavity of eight oxygen atoms.
Article PDF
Similar content being viewed by others

Avoid common mistakes on your manuscript.
References and Notes
Part 15 in Molecular Inclusion in Functionalized Macrocycles. For part 14 see Ref. 6.
D. J. Cram:Angew. Chem. Int. Ed. Engl. 25, 1039 (1986) and references therein.
A. Arduini, A. Pochini, S. Reverberi, and R. Ungaro:J. Chem. Soc. Chem. Commun. 981 (1984).
M. A. McKervey, E. M. Seward, G. Ferguson, B. Ruhl, and S. J. Harris:J. Chem. Soc. Chem. Commun. 388 (1985); S. K. Chang and I. Cho,J. Chem. Soc. Perkin Trans. 1, 211 (1986).
A. Arduini, A. Pochini, S. Reverberi, R. Ungaro, G. D. Andreetti, and F. Ugozzoli:Tetrahedron,42, 2089, (1986).
Preliminary communication: G. Calestani, F. Ugozzoli, A. Arduini, E. Ghidini, and R. Ungaro:J. Chem. Soc. Chem. Commun. 344 (1987).
For the nomenclature of calixarenes and their derivatives, see G. D. Gutsche:Top. Curr. Chem. 123, 1 (1984).
N. L. Drake, C. H. Eaker, and W. Shenk:J. Chem. Soc. 70, 677 (1948).
D. Belletti, F. Ugozzoli, A. Cantoni, and G. Pasquinelli: Centro di Studio per la Strutturistica Diffrattometrica del CNR, Parma, Italy, Internal Reports 1/79, 2/79, 3/79 (1979).
M. S. Lehman and F. K. Larsen:Acta Crystallogr. A30, 80 (1974).
F. Ugozzoli:Computers & Chemistry 11, 109 (1987).
G. M. Sheldrick: SHELX76.Program for Crystal Structure Determination. Univ. of Cambridge, England (1976).
Cf. U. Takaki, T. E. Hogen-Esh, and J. Smid:J. Am. Chem. Soc. 93, 6760 (1971).
J. Smid and R. Sinta:Top. Curr. Chem. 121, 105 (1984).
V. Gutmann:Coordination Chemistry Review 18, 225 (1976).
We thank a referee for suggesting that the presence in ligand (2) of ether-amide binding groups could enhance its complexing ability toward alkaline-earth cations, by analogy with calcium-selective ether-amide podands (cf. W. E. Morfet al.: Progress in Macrocyclic Chemistry (R. M. Izatt and J. J. Christensen, Eds.) Wiley-Interscience 1979, p. 1).1H titration experiments, similar to that described in this paper, show that ligand (2) is indeed able to complex Ca(SCN)2 in CDCl3 with a 1∶1 stoichiometry. However, since we do not have the overall picture of the association constants, we cannot discuss the problem of monovalent/divalent cation selectivity of our ligand (2).
Author information
Authors and Affiliations
Additional information
Supplementary Data relating to this article are deposited with the British Library as supplementary publication No. SUP 82059 (57 pages).
Rights and permissions
About this article
Cite this article
Arduini, A., Ghidini, E., Pochini, A. et al. p-t-Butylcalix[4]arene tetra-acetamide: a new strong receptor for alkali cations [1]. Journal of Inclusion Phenomena 6, 119–134 (1988). https://doi.org/10.1007/BF00663045
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00663045