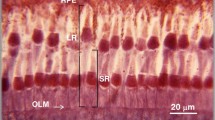
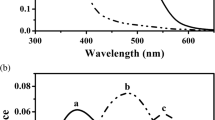
Summary
Spectrophotometric measurements of photoreceptors 1–6 in the blowfly demonstrate that rhodopsin undergoes a continuous renewal. This involves, in the dark, the slow degradation of rhodopsin whereas metarhodopsin is degraded at a much faster rate. The effect of light is to reduce the rate at which metarhodopsin is degraded, i.e. the rate is inversely related to the intensity of the light. Rhodopsin synthesis is dependent on the presence of 11-cis retinal which is formed via a photoreaction from all-trans retinal resulting from the breakdown of rhodopsin and/or metarhodopsin: the biosynthesis of rhodopsin is therefore a light dependent process. Light of the blue/violet spectral range was found to mediate the isomerization of all-trans retinal into the 11-cis form. It is proposed that this stereospecificity is the result of all-trans retinal being bound to a protein. On the basis of the results a visual pigment cycle is proposed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Autram H (1981) Light and dark adaptation in invertebrates. In: Autrum H (ed) Comparative physiology and evolution of vision in invertebrates. (Handbook of sensory physiology, vol VII/6C) Springer, Berlin Heidelberg New York, pp 1–91
Bernard DG (1983) Dark-processes following photoconversion of butterfly rhodopsins. Biophys Struct Mech 9:277–286
Blest AD (1978) The rapid synthesis and destruction of photoreceptor membrane by a dinopid spider: a daily cycle. Proc R Soc London Ser B 200:463–483
Blest AD (1980) Photoreceptor membrane turnover in arthropods: comparative studies of breakdown processes and their implications. In: Williams TP, Baker BN (eds) The effect of constant light on visual processes. Plenum, New York London, pp 217–245
Boschek BC, Hamdorf K (1976) Rhodopsin particles in the photoreceptor membrane of an insect. Z Naturforsch 31c:763
Brown PK, Brown PS (1958) Visual pigments of the octopus and cuttle fish. Nature 182:1288–1290
Brown PK, White RH (1972) Rhodopsin of larval mosquito. J Gen Physiol 59:401–414
Burnel M, Mahler HR, Moore WJ (1970) Protein synthesis in visual cells ofLimulus. J Neurochem 17:1493–1499
Eguchi E (1965) Rhabdom structure and receptor potentials in single crayfish retinular cells. J Cell Comp Physiol 66:411–430
Eguchi E, Waterman TH (1966) Fine structure patterns in crustacean rhabdoms. In: Bernhard CG (ed) Functional organization of the compound eye. Wenner Green Internatl Symp Series 7. Pergamon Press, Oxford, pp 105–124
Eguchi E, Waterman TH (1967) Changes in retinal fine structure induced in the crabLibinia by light and dark adaptation. Z Zellforsch 79:209–229
Goldman LJ, Barnes SN, Goldsmith TH (1975) Microspectrophotometry of rhodopsin and metarhodopsin in the mothGalleria. J Gen Physiol 66:383–404
Goldsmith TH (1958) The visual system of the honeybee. Proc Natl Acad Sci USA 44:123–126
Goldsmith TH, Bruno M (1973) Behavior of rhodopsin and metarhodopsin in isolated rhabdoms of crabs and lobster. In: Langer H (ed) Biochemistry and physiology of visual pigments. Springer, Berlin Heidelberg New York, pp 147–153
Hamdorf K (1979) The physiology of invertebrate visual pigments. In: Autrum H (ed) Invertebrate photoreceptors. (Handbook of sensory physiol, vol VII/6A) Springer, Berlin Heidelberg New York, pp 145–224
Hamdorf K, Schwemer J (1975) Photoregeneration and the adaptation process in insect photoreceptors. In: Snyder AW, Menzel R (eds) Photoreceptor optics. Springer, Berlin Heidelberg New York, pp 263–289
Hamdorf K, Schwemer J, Gogala M (1971a) Insect visual pigment sensitive to ultraviolet light. Nature 231:458–459
Hamdorf K, Gogala M, Schwemer J (1971b) Beschleunigung der Dunkeladaptation eines UV-Rezeptors durch sichtbare Strahlung. Z Vergl Physiol 75:189–199
Hamdorf K, Paulsen R, Schwemer J, Täuber U (1972) Photoreconversion of invertebrate visual pigments. In: Wehner R (ed) Information processing in the visual system of arthropods. Springer, Berlin Heidelberg New York, pp 97–108
Hamdorf K, Paulsen R, Schwemer J (1973) Photoregeneration and sensitivity control of photoreceptors of invertebrates. In: Langer H (ed) Biochemistry and physiology of visual pigments. Springer, Berlin Heidelberg New York, pp 155–166
Hara T, Hara R (1965) New photosensitive pigment found in the retina of the squidOmmastrephes. Nature 206:1331–1334
Hara T, Hara R (1972) Cephalopod retinochrome. In: Dartnall HJA (ed) Photochemistry of vision. (Handbook of sensory physiology, vol VII/1) Springer, Berlin Heidelberg New York, pp 720–746
Harris WA, Ready DF, Lipson ED, Hudspeth AJ, Stark WS (1977) Vitamin A deprivation andDrosophila photopigments. Nature 266:648–650
Hillman P, Hochstein S, Minke B (1983) Transduction in invertebrate photoreceptors: role of pigment bistability. Physiol Rev 63:668–772
Hubbard R, St George RCC (1958) The rhodopsin system of the squid. J Gen Physiol 41:501–528
Kropf A, Brown PK, Hubbard (1959) Lumi- and meta-rhodopsin of squid and octopus. Nature 183:446–448
Paulsen R, Schwemer J (1979) Vitamin A-deficiency reduces the concentration of visual pigment protein within blowfly photoreceptor membranes. Biochim Biophys Acta 557:385–390
Paulsen R, Schwemer J (1983) Biogenesis of blowfly photoreceptor membranes is regulated by 11-cis retinal. Eur J Biochem 137:609–614
Pepe IM, Schwemer J, Paulsen R (1982) Characteristics of retinal-binding proteins from the honeybee retina. Vision Res 22:775–781
Perrelet A (1972) Protein synthesis in the visual cells of the honeybee drone as studied with electron microscope radioautography. J Cell Biol 55:595–605
Schwemer J (1969) Der Sehfarbstoff vonEledone moschata und seine Umsetzungen in der lebenden Netzhaut. Z Vergl Physiol 62:121–152
Schwemer J (1983) Pathways of visual pigment regeneration in fly photoreceptor cells. Biophys Struct Mech 9:287–298
Schwemer J, Gogala M, Hamdorf K (1971) Der UV-Sehfarbstoff der Insekten: Photochemie in vitro und in vivo. Z Vergl Physiol 75:174–188
Schwemer J, Henning U (in press) Morphological correlates of visual pigment turnover in fly photoreceptors. Cell Tissue Res
Schwemer J, Pepe IM, Paulsen R, Cugnoli C (1984) Lightactivatedtrans-cis isomerization of retinal by a protein from honeybee retina. J Comp Physiol A 154:549–554
Seki T, Hara R, Hara T (1980) Reconstitution of squid rhodopsin in rhabdomal membranes. Photochem Photobiol 32:469–479
Stavenga DG, Schwemer J (in press) Visual pigments of invertebrates. In: Ali MA (ed) Photoreception and vision in invertebrates. Plenum Press, New York
Stavenga DG, Zantema A, Kuiper JW (1973) Rhodopsin processes and the function of the pupil mechanism in flies. In: Langer H (ed) Biochemistry and physiology of visual pigments. Springer, Berlin Heidelberg New York, pp 175–180
Stein PJ, Brammer JD, Ostroy SE (1979) Renewal of opsin in the photoreceptor cells of the mosquito. J Gen Physiol 74:565–582
Tuurala O, Lehtinen A (1974) Inkorporierung des tritiummarkierten Leucins in die Sehzellen vonOniscus asellus L. (Isopoda, Oniscoidea). Ann Zool Fennici 11:135–140
White RH (1964) The effect of light upon the ultrastructure of the mosquito eye. Am Zool 4:433
White RH (1967) The effect of light deprivation upon the ultrastructure of larval mosquito ey. II. The rhabdom. J Exp Zool 166:405–425
White RH (1968) The effect of light and light deprivation upon the ultrastructure of the larval mosquito eye. III. Multivesicular bodies and protein uptake. J Exp Zool 169:261–278
Williams DS (1982) Rhabdom size and photoreceptor membrane turnover in a muscoid fly. Cell Tissue Res 226:629–639
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwemer, J. Renewal of visual pigment in photoreceptors of the blowfly. J. Comp. Physiol. 154, 535–547 (1984). https://doi.org/10.1007/BF00610167
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00610167