Summary
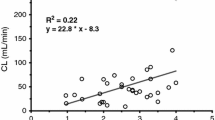
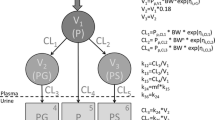
The pharmacokinetics and metabolism of lignocaine in premature neonates was studied after subcutaneous administration. The collection of serial urine together with a limited number of blood samples from neonates enabled simultaneous computer fitting of data to a pharmacokinetic model. The disposition kinetics of lignocaine in four neonates were compared with similar data reported for adults. Neonates had prolonged t1/2 (neonate mean: 3.16 h; adult mean: 1.80 h), and an increased total volume of distribution (neonate mean: 2.75 l/kg; adult mean: 1.11 l/kg) compared with adults. Total plasma clearance (Cltp) normalised on body weight showed no significant difference between neonates (mean: 0.610 l/h/kg) and adults (mean: 0.550 l/h/kg). The urinary excretion of lignocaine and several of its metabolites was studied in 8 neonates and 11 adults. Neonates were shown to excrete much more unchanged lignocaine (mean: 19.67%) compared with adults (mean: 4.27%) and the proportion of the dose excreted as 4-hydroxyxylidine is considerably reduced in neonates (neonate mean: 8.89%; adult mean: 63.78%). The use of the two pharmacokinetic parameters, t1/2 and Cltp, as indices of drug elimination ability are discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Boxenbaum, H.G., Riegelman, S., Elashoff, R.M.: Statistical estimations in pharmacokinetics. J. Pharmacokin. Biopharm.2, 123–148 (1974)
Brown, W.U., Bell, G.C., Lurie, A.O., Weiss, J.B., Scanlon, J.W., Alper, M.H.: Newborn blood levels of lignocaine and mepivicaine in the first postnatal day following maternal epidural anesthesia. Anesthesiology12, 698–707 (1975)
Dixon, W.J., Massey, F.J.: Introduction to statistical analysis, pp. 349–351. New York: McGraw Hill Inc. 1969
Garrettson, L.K., Procknal, J.A., Levy, G.: Fetal acquisition and neonatal elimination of a large amount of salicylate. Clin. Pharmacol. Ther.17, 98–103 (1975)
Gibaldi, M., Perrier, D.: Pharmacokinetics, Chap. 9. Marcel Dekker Inc., New York: Marcel Dekker Inc. 1975
Goldstein, A.: Biostatistics: An introductory text, pp. 55–58. New York: The Macmillan Co. 1964
Keenaghan, J.B., Boyes, R.N.: The tissue distribution, metabolism and excretion of lignocaine in rats, guinea pigs, dogs and man. J. Pharmacol. Exp. Ther.180, 454–463 (1972)
Levy, G., Khanna, N.N., Soda, D.M., Tsuki, O., Stern, L.: Pharmacokinetics of acetaminophen in the human neonate: Formation of acetaminophen glucuronide and sulphate in relation to plasma bilirubin concentration and d-glucaric acid excretion. Pediatrics55, 818–825 (1975)
Loggie, J.M.H., Kleinman, L.I., VanMaanen, E.F.: Renal function and diuretic therapy in infants and children Part I. J. Pediat.86, 485–496 (1975)
Meffin, P., Long, G.J., Thomas, J.: Clearance and metabolism of mepivicaine in the human neonate. Clin. Pharmacol. Ther.14, 218–225 (1973)
Metzler, C.: NONLIN Program, Division of Mathematical Services Kalamazoo, Mich.: The Upjohn Company 1969
Miller, R.P., Roberts, R.J., Fischer, L.J.: Acetaminophen elimination kinetics in neonates, children and adults. Clin. Pharmacol. Ther.19, 284–294 (1976)
Morselli, P.: Clinical pharmacokinetics in neonates. Clin. Pharmacokinetics1, 81–98 (1976)
Morselli, P., Principi, N., Tognoni, G., Reali, E., Belvedere, G., Standen, S.M., Sereni, F.: Diazepam elimination in premature and full term infants, and children. J. Perinat. Med.1, 133–141 (1973)
Nation, R.L., Triggs, E.J., Selig, M.: Gas chromatographic method for the quantitative determination of lignocaine and its metabolite monoethylglycinexylidide in plasma. J. Chromatogr.116, 190–193 (1976)
Perrier, D., Gibaldi, M.: Clearance and biological half-life as indices of intrinsic hepatic metabolism. J. Pharmacol. Exp. Ther.191, 17–24 (1974)
Rowe, F.M., Bannister, S.H., Seth, R.R., Storey, R.C.: Derivatives of m-xylenols. Part 1. Intermediate products from m-5-xylenol (5-hydroxy-1,3-dimethylbenzene). J. Soc. Chem. Ind.49, 469–473T. (1930)
Rowland, M., Thompson, P. D., Guichard, A., Melmon, K. L.: Disposition kinetics of lignocaine. Ann. N. Y. Acad. Sci.179, 383–398 (1971)
Thomas, J., Morgan, D., Vine, J.: Metabolism of etidocaine in man. Xenobiotica6, 39–48 (1976)
Tucker, G.T., Boyes, R.N., Bridenbaugh, P.O., Moore, D.C.: Binding of anilide type local anesthetics in human plasma; I. Relationship between binding, physicochemical properties and anesthetic activity. Anesthesiology33, 287–303 (1970 a)
Tucker, G.T., Boyes, R.N., Bridenbaugh, P.O., Moore, D.C.: Binding of anilide type local anesthetics in human plasma; II. Implications in vivo, with special reference to transplacental distribution. Anesthesiology33, 304–314 (1970 b)
Yaffe, S.J., Juchau, M.R.: Perinatal Pharmacology. Ann. Rev. Pharmacol.14, 219–238 (1974)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mihaly, G.W., Moore, R.G., Thomas, J. et al. The pharmacokinetics and metabolism of the anilide local anaesthetics in neonates. Eur J Clin Pharmacol 13, 143–152 (1978). https://doi.org/10.1007/BF00609759
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609759