Summary
-
1.
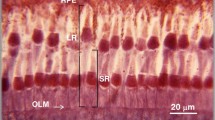
Intracellular recordings were made of receptor responses in the central region of the compound eyes of the locusts,Valanga andLocusta. The animals were maintained on their usual daily light cycle, and recordings were made at times known from previous anatomical studies to coincide with changes in ommatidial structure (Tunstall and Horridge 1967; Horridge et al. 1981; Williams 1982a). Anatomical checks were made of the areas of the retina from which recordings had been made (Fig. 1).
-
2.
Angular acceptance at 50% sensitivity inValanga andLocusta respectively increased from 1.7° and 1.9° when light-adapted during the day, to 2.7° and 2.8° when dark-adapted for 10–15 min after ‘dusk’, to 4.7° and 4.9° in a fully night-adapted state. It then decreased to 2.05° and 1.9° when light-adapted for 2 h after ‘dawn’, and increased to 2.8° and 2.9° after a further 20 min dark adaptation (Table 1). Dark-adapted values were measured by exposing the cells to very dim light and counting the quantum bumps (Fig. 3), thus ensuring that there were no light-adaptive effects from the stimulus.
-
3.
Sensitivity to an extended source increased in both species by at least 1 log unit during the first 1–3 h after ‘dusk’ before reaching its maximum (Fig. 6). The last 0.6 log units of this increment (3.9-fold increase) is attributed to the enlargement of the field stop and rhabdom. After 2 h light from ‘dawn’, which induces diminution of the rhabdom and field stop to their day sizes, followed by 20 min of dark adaptation, cells were 3.8 times (data fromValanga andLocusta grouped) less sensitive to the extended source than they were at night.
-
4.
The increased light capture from the environment, resulting from opening up the rhabdom's acceptance at night, is obtained at the expense of spatial acuity. At night, the angular acceptance at 50% sensitivity becomes more than twice the interommatidial angle (Δϕ) (Fig. 8). As a result, discrimination of two points spaced at 2Δϕ, which is possible during the day, becomes impossible at night.
-
5.
No change of spectral sensitivity (Fig. 4), bump latency (Fig. 7), or sensitivity to a point source on-axis (Fig. 5) was detected between the night and day states. The last indicates that the absorption efficiency of the rhabdom is constant, and the Airy disc is smaller than the day-state field stop. Together these findings indicate that the new photoreceptor membrane assembled at dusk does not differ fundamentally from that maintained during the day. Changes in sensitivity are therefore achieved by changing the amount of exposed receptor membrane rather than the nature of the membrane.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barlow RB, Chamberlain SC, Levison SZ (1980a)Limulus brain modulates the structure and function of the lateral eyes. Science 210:1037–1039
Burtt ET, Catton WT (1954) Visual perception of movement in the locust. J Physiol (Lond) 125:566–580
Götz KG (1964) Optomotorische Untersuchung des visuellen Systems einiger Augenmutanten der FruchtfliegeDrosophila. Kybernetik 2:77–92
Hamdorf K, Kirschfeld K (1980) “Prebumps”: evidence for double-hits at functional subunits in a rhabdomeric photoreceptor. Z Naturforsch 35c:173–174
Horridge GA (1978) The separation of visual axes in apposition compound eyes. Philos Trans R Soc Lond [Biol] 285:1–59
Horridge GA, Barnard PBI (1965) Movement of palisade in locust retinula cells when illuminated. Q J Microsc Sci 106:131–135
Horridge GA, Duniec J, Marcelja L (1981) A 24-hour cycle in single locust and mantis photoreceptors. J Exp Biol 91:307–322
Jahn TL, Wulff VJ (1943) Electrical aspects of a diurnal rhythm in the eye ofDytiscus fasciventris. Physiol Zool 16:101–109
Kaplan E, Barlow RB (1980) Circadian clock inLimulus brain increases response and decreases noise of retinal photoreceptors. Nature 286:393–395
Kirschfeld K (1976) The resolution of lens and compound eyes. In: Zettler F, Weiler R (eds) Neural principles in vision. Springer, Berlin Heidelberg New York, pp 354–372
Kuwabara T (1970) Retinal recovery from exposure to light. Am J Ophthalmol 70:187–198
Laughlin SB (1975) Receptor function in the apposition eye — an electrophysiological approach. In: Snyder AW, Menzel R (eds) Photoreceptor optics. Springer, Berlin Heidelberg New York, pp 479–798
Laughlin S (1980) Neural principles in the peripheral visual systems of invertebrates. In: Autrum H (ed) Vision in invertebrates. Springer, Berlin Heidelberg New York (Handbook of sensory physiology, vol VII/6B, pp 133–280)
Laughlin S, Blest AD, Stowe S (1980) The sensitivity of receptors in the posterior median eye of the nocturnal spider,Dinopis. J Comp Physiol 141:53–65
Leggett LMW, Stavenga DG (1981) Diurnal changes in angular sensitivity of crab photoreceptors. J Comp Physiol 144:99–109
Lillywhite PG (1977) Single photon signals and transduction in an insect eye. J Comp Physiol 122:189–200
Lillywhite PG (1978) Coupling between locust photoreceptors revealed by a study of quantum bumps. J Comp Physiol 125:13–28
Meyer-Rochow VB, Horridge GA (1975) The eye ofAnoplognathus (Coleoptera, Scarabaeidae). Proc R Soc Lond [Biol] 188:1–30
Rossel S (1979) Regional differences in photoreceptor performance in the eye of the praying mantis. J Comp Physiol 131:95–112
Scholes JH (1964) Discrete subthreshold potentials from the dimly lit insect eye. Nature 202:572–573
Shaw SR (1969) Interreceptor coupling in ommatidia of drone honeybee and locust compound eyes. Vision Res 9:999–1029
Snyder AW (1977) Acuity of compound eyes: physical limitations and design. J Comp Physiol 116:161–182
Snyder AW (1979) The physics of vision in compound eyes. In: Autrum H (ed) Vision in invertebrates. Springer, Berlin Heidelberg New York (Handbook of sensory physiology, vol VII/6A, pp 225–313)
Stavenga DG (1979) Pseudopupils of compound eyes. In: Autrum H (ed) Vision in invertebrates. Springer, Berlin Heidelberg New York (Handbook of sensory physiology, vol VII/6A, pp 357–439)
Stowe S (1980a) Spectral sensitivity and retinal pigment movement in the crabLeptograpsus variegatus (Fabricius). J Exp Biol 87:73–98
Stowe S (1980b) Rapid synthesis of photoreceptor membrane and assembly of new microvilli in a crab at dusk. Cell Tissue Res 211:419–440
Swihart SL (1963) The electroretinogram ofHeliconius erato (Lepidoptera). Zoologica 48:155–166
Tunstall J, Horridge GA (1967) Electrophysiological investigation of the optics of the locust retina. Z Vergl Physiol 55:167–182
Walcott B (1971) Unit studies on receptor movement in the retina ofLethocerus (Belostomatidae, Hemiptera). Z Vergl Physiol 74:1–16
White RH, Lord E (1975) Diminution and enlargement of the mosquito rhabdom in light and darkness. J Gen Physiol 65:583–598
Williams DS (1982a) Ommatidial structure in relation to photoreceptor membrane turnover in the locust. Cell Tissue Res 225:595–617
Williams DS (1982b) Photoreceptor membrane shedding and assembly can be initiated locally within an insect retina. Science 218:898–900
Wilson M (1975) Angular sensitivity of light and dark adapted locust retinula cells. J Comp Physiol 97:323–328
Wilson M, Garrard P, McGinness S (1978) The unit structure of the locust compound eye. Cell Tissue Res 195:205–226
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Williams, D.S. Changes of photoreceptor performance associated with the daily turnover of photoreceptor membrane in locusts. J. Comp. Physiol. 150, 509–519 (1983). https://doi.org/10.1007/BF00609577
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609577