Abstract
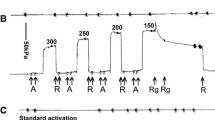
Force-velocity relations, rate of ATP turnover (JATP), and phosphorylation of the 20,000 D myosin light chains (LC20) were measured in chemically skinned guinea pigTaenia coli. Relative LC20 phosphorylation at 3.2 mM MgATP was 17% in relaxed tissues at pCa 9, and increased with force at increasing [Ca2+] to a maximum of 67% at pCa 4.5. Force at pCa 4.5 was dependent on the MgATP concentration with a half-maximal response at about 0.1 mM. At 0.1 mM MgATP LC20 phosphorylation at pCa 4.5 was 38%. Both JATP and the maximal shortening velocity (V max) were reduced in 0.1 mM MgATP, to 32% and 43%, respectively, of their values at 3.2 mM MgATP. Low-MgATP thus inhibits both LC20 phosphorylation and the extent and rate of cross-bridge interaction. High levels of LC20 phosphorylation, independent of Ca2+ and MgATP concentrations, were obtained by treatment with ATP-γ-S. Maximal force at 3.2 mM MgATP after LC20 thiophosphorylation was unchanged, whereas halfmaximal force occurred at 0.065 mM MgATP after thiophosphrylation, compared to 0.13 mM after activation by Ca2+. The contraction in thiophosphorylated preparations at low-MgATP (0.1 mM) was associated with submaximalV max (60%) and JATP (27%). The results show that LC20 phosphorylation is correlated with the degree of force development in the Ca2+ activated contraction, both when Ca2+ and MgATP concentrations are varied. The reduced force and rate of crossbridge turnover in lowMgATP are however primarily mediated by an influence of MgATP on the cross-bridge cycle, which is separate from the effect on LC20 phosphorylation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adelstein RS, Klee CB (1981) Purification and characterization of smooth muscle myosin light chain kinase. J Biol Chem 256:7501–7509
Arner A (1982) Mechanical characteristics of chemically skinned guinea pigTaeni coli. Pflügers Arch 395:277–284
Arner A (1983) Force-velocity relation in chemically skinned rat portal vein. Effects of Ca2+ and Mg2+. Pflügers Arch 397:6–12
Arner A, Hellstrand P (1983) Activation of contraction and ATPase activity in intact and chemically skinned smooth muscle of rat portal vein. Circ Res 53:695–702
Arner A, Hellstrand P (1985) Effects of calcium and substrate on force-velocity relation and energy turnover in skinned smooth muscle of the guinea-pig. J Physiol 360:347–365
Cassidy P, Hoar PE, Kerrick WGL (1979) Irreversible thiophosphorylation and activation of tension in functionally skinned rabbit ileum strips by [35S]ATP-γ-S. J Biol Chem 254:11148–11153
Cooke R, Bialek W (1979) Contraction of glycerinated muscle fibers as a function of the ATP concentration. Biophys J 28:241–258
Dillon PF, Aksoy MO, Driska SP, Murphy RA (1981) Myosin phosphorylation and the cross-bridge cycle in arterial smooth muscle. Science 211:495–497
DiSalvo J, Miller J, Blumenthal D, Stull JT (1981) Isolation and enzymatic properties of myosin light chain kinase from vascular smooth muscle. Biophys J 33:276a
Driska SP, Aksoy MO, Murphy RA (1981) Myosin light chain phosphorylation associated with contraction in arterial smooth muscle. Am J Physiol 240:C222-C233
Fabiato A (1981) Myoplasmic free calcium concentration reached during twitch of an intact isolated cardiac cell and during calcium-induced release of calcium from the sarcoplasmic reticulum of a skinned cardiac cell from the adult rat or rabbit ventricle. J Gen Physiol 78:457–497
Fabiato A, Fabiato F (1979) Calculator programs for computing the composition of the solutions containing multiple metals and ligands used for experiments in skinned muscle cells. J Physiol (Paris) 75:463–505
Ferenczi MA, Goldman YE, Simmons RM (1984) The dependence of force and shortening velocity on substrate concencentration in skinned muscle fibres fromRana temporaria. J Physiol 350: 519–543
Fletcher R, Rowell JD (1963) A rapidly convergent descent method for minimization. Computer J 6:163–168
Gagelmann M, Güth K (1985) Force generated by non-cycling cross-bridges at low ionic strength in skinned smooth muscle fromTaenia coli. Pflügers Arch 403:210–214
Gagelmann M, Rüegg JC, DiSalvo J (1984) Phosphorylation of the myosin light chains and satellite proteins in detergent-skinned arterial smooth muscle. Biochem Biophys Res Commun 120: 933–938
Güth K, Mrwa U (1982) Maximum force is generated in chemically skinnedTaenia coli at lower Ca2+ concentrations than maximum ATPase activity. Pflügers Arch 394:R44
Haeberle JR, Hott JW, Hathaway DR (1984) Pseudophosphorylation of the smooth muscle 20,000 dalton myosin light chain. Biochem Biophys Acta 790:78–86
Haeslip RJ, Chacko S (1985) Effects of Ca2+ and Mg2+ on the actomyosin adenosine-5′-triphosphatase of stably phosphorylated gizzard myosin. Biochemistry 24:2731–2736
Hellstrand P, Johansson B (1979) Analysis of the length response to a force step in smooth muscle from rabbit urinary bladder. Acta Physiol Scand 106:221–238
Hellstrand P, Paul RJ (1982) Vascular smooth muscle: Relations between energy metabolism and mechanics. In: Crass MF III, Barnes CD (ed) Vascular smooth muscle: metabolic, ionic, and contractile mechanisms, Cht. 1. Academic Press, New York, p1
Hill AV (1938) The heat of shortening and the dynamic constants of muscle. Proc Roy Soc B 126:136–195
Hoar PE, Kerrick WGL, Cassidy PS (1979) Chicken gizzard: relation between calcium-activated phosphorylation and contraction. Science 204:503–506
Kushmerick MJ, Krasner B (1982) Force and ATPase rate in skinned skeletal muscle fibers. Fed Proc 41:2232–2237
Ledvora RF, Barany K, VanderMeulen DL, Barron JT, Barany M (1983) Two dimension gel electrophoretic analysis of myosin light chain phosphorylation in contracting and resting vascular smooth muscle. Fed Proc 42:736
Marston SB (1983) Myosin and actomyosin ATPase: kinetics. In: Stephens NL (ed) Biochemistry of smooth muscle, Vol 1, Cht 4. CRC Press, Boca Raton, FL, p 167
Paul RJ, Doermann G, Zeugner C, Rüegg JC (1983) The dependence of unloaded shortening velocity on Ca2+, calmodulin and duration of contraction in “chemically-skinned” smooth muscle. Circ Res 53:342–351
Peterson JW (1980) Vanadate ion inhibits actomyosin interaction in chemically skinned vascular smooth muscle. Biochem Biophys Res Commun 95:1846–1853
Peterson JW (1982) Simple model of smooth muscle myosin phosphorylation and dephosphorylation as rate-limiting mechanism. Biophys J 37:453–459
Rüegg JC (1971) Smooth muscle tone. Physiol Rev 51:201–247
Sherry JMF, Gorecka A, Aksoy MO, Dabrowska R, Hartshorne DJ (1978) Roles of calcium and phosphorylation in the regulation of the activity of gizzard myosin. Biochemistry 17:4411–4418
Takashi R, Putnam S (1979) A fluorimetric method for continuously assaying ATPase: application to small specimens of glycerolextracted muscle fibres. Anal Biochem 92:375–382
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hellstrand, P., Arner, A. Myosin light chain phosphorylation and the cross-bridge cycle at low substrate concentration in chemically skinned guinea pigTaenia coli . Pflugers Arch. 405, 323–328 (1985). https://doi.org/10.1007/BF00595684
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00595684