Abstract
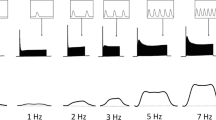
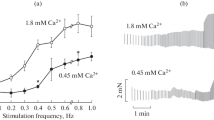
The rate of resting heat production of quiescent rat left ventricular papillary muscles was measured myothermically. The effects of contractile activity, stretch, oxygen partial pressure, temperature, amino acids and time were examined. The rate of basal heat production was the same throughout the day whether or not muscles contracted isotonically under a small pre-load. Passive stretch increased the rate of resting heat production; the stretch-induced increment was highly variable from muscle to muscle. The resting heat rate per se was only moderately sensitive to oxygen partial pressure and temperature, and was insensitive to the presence of amino acids in the bathing medium. The stretch-induced increase in resting heat rate was independent of these three factors. The rate of resting heat production declined exponentially with time to reach a plateau about 4 h following cardiectomy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ballard FJ (1977) Intracellular protein degradation. Essays Biochem 13:1–37
Baños G, Daniel PM, Moorhouse SR, Pratt OE, Wilson PA (1978) The influx of amino acids into the heart of the rat. J Physiol 280:471–486
Burns AH, Reddy WJ (1978) Amino acid stimulation of oxygen and substrate utilization by cardiac myocytes. Am J Physiol 235:E461-E466
Chapman JB (1983) Heat production. In: Drake-Holland AJ, Noble MIM (eds) Cardiac metabolism. Wiley, Chichester
Chapman JB, Gibbs CL (1974) The effect of metabolic substrate on mechanical activity and heat production in papillary muscle. Cardiovasc Res 8:656–667
Chapman JB, Gibbs CL, Loiselle DS (1977) Simultaneous heat and fluorescence changes in cardiac muscle at high rates of energy expenditure: effect of caffeine and isoprenaline. J Mol Cell Cardiol 9:715–732
Chapman JB, Gibbs CL, Loiselle DS (1982) Myothermic, polarographic and fluorometric data from mammalian muscles: correlations and an approach to a biochemical synthesis. Fed Proc 41:176–184
Coleman HN, Sonneblick EH, Braunwald E (1971) Mechanism of norepinephrine-induced stimulation of myocardial oxygen consumption. Am J Physiol 221:778–783
Cranefield PF, Greenspan K (1960) The rate of oxygen consumption of quiescent cardiac muscle. J Gen Physiol 44:235–249
Delbridge LM, Loiselle DS (1981) An ultrastructural investigation into the size dependency of contractility of isolated cardiac muscle. Cardiovasc Res 15:21–27
Earl CA, Laurent GJ, Everett AW, Bonnin CM, Sparrow MP (1978) Turnover rates of muscle protein in cardiac and skeletal muscles of dog, fowl, rat and mouse: turnover rate related to muscle function. Aust J Exp Biol Med 56:265–277
Everett AW, Sparrow MP, Taylor RR (1979) Early changes in myocardial protein synthesis in vivo in response to right ventricular pressure overload in the dog. J Mol Cell Cardiol 11:1253–1263
Fuhrman GJ, Fuhrman FA, Field J (1950) Metabolism of rat heart slices, with special reference to effects of temperature and anoxia. Am J Physiol 163:642–647
Gibbs GL (1978) Cardiac energetics. Physiol Rev 58:174–254
Gibbs CL (1982) Modification of the physiological determinants of cardiac energy expenditure by pharmacological agents. Pharmacol Ther 18:133–157
Gibbs CL, Chapman JB (1979a) Cardiac heat production. Annu Rev Physiol 41:507–519
Gibbs CL, Chapman JB (1979b) Cardiac energetics. In: Berne RM, Sperelakis N (eds) The cardiovascular system. Handbook of physiology, ch 22. Am Physiol Soc, Bethesda MD, pp 775–804
Gibbs CL, Gibson WR (1969) Effect of ouabain on the energy output of rabbit cardiac muscle. Circ Res 24:951–967
Gibbs CL, Gibson WR (1972) Isoprenaline, propranolol and the energy output of rabbit cardiac muscle. Cardiovasc Res 6:508–515
Gibbs CL, Kotsanas G (1983) The metabolism of quiescent rabbit myocardium. Proc Aust Physiol Pharmacol Soc 14:18
Gibbs CL, Mommaerts WFMH, Ricchiuti NV (1967) Energetics of cardiac contractions. J Physiol 191:25–46
Gibbs CL, Papadoyannis DE, Drake AJ, Noble MIM (1980) Oxygen consumption of the nonworking and potassium chloride-arrested dog heart. Circ Res 47:408–417
Greenspan K, Cranefield PF (1963) Influence of some factors on oxygen uptake of canine cardiac Purkinje fibres. Am J Physiol 24:5–8
Hill AV (1928) The diffusion of oxygen and lactic acid through tissues. Proc Roy Soc London Series B 104:39–96
Hjalmarson A, Isaksson O (1972) In vitro work load and rat heart metabolism. I. Effect on protein synthesis. Acta Physiol Scand 86:126–144
Johnstone RM (1979) Electrogenic amino acid transport. Can J Physiol Pharmacol 57:1–15
Lee KS (1960) The relationship of the oxygen consumption to the contraction of the cat papillary muscle. J Physiol 151:186–201
Lesch M, Gorlin R, Sonnenblick EH (1970) Myocardial amino acid transport in the isolated rabbit right ventricular papillary muscle. Circ Res 27:445–460
Lochner W, Arnold G, Muller-Ruchholtz ER (1968) Metabolism of the artificially arrested heart and of the gas-perfused heart. Am J Cardiol 22:299–311
Loiselle DS (1982) Stretch-induced increase in resting metabolism of isolated papillary muscle. Biophys J 38:185–195
Loiselle DS (1985) The effect of temperature on the basal metabolism of cardiac muscle. Pflügers Arch 405:163–169
Loiselle DS, Gibbs CL (1979) Species differences in cardiac energetics. Am J Physiol 237:H90-H98
Loiselle DS, Gibbs CL (1983) Factors affecting the metabolism of resting rabbit papillary muscle. Pflügers Arch 396:285–291
Millward DJ, Garlick PJ, James WPT, Sender P, Waterlow JC (1976) Protein turnover. In: Cole DJA, Boorman KN, Buttery PJ, Lewis D, Neale RJ, Swan H (eds) Protein metabolism and nutrition; proceedings. Butterworths, London, pp 49–69
Morgan HE, Kira Y, Gordon EE, Kochel PJ (1983) Mechanical factors determining rates of protein synthesis and degradation. J Mol Cell Cardiol, suppl 2 15:19
Page E, McCallister LP, Power B (1971) Stereological measurement of cardiac ultrastructure implicated in excitation-contraction coupling. Proc Natl Acad Sci USA 68:1465–1466
Penpargkul S, Scheuer J (1969) Metabolic comparisons between hearts arrested by calcium deprivation or potassium excess. Am J Physiol 217:1405–1412
Peterson MB, Lesch M (1972) Protein synthesis and amino acid transport in the isolated rabbit right ventricular papillary muscle. Circ Res 31:317–327
Pool PE, Sonnenblick EW (1967) The mechanochemistry of cardiac muscle. I. The isometric contraction. J Gen Physiol 50:951–965
Ray AA (1982) (ed) SAS user's guide: Statistics. SAS Institute Inc, Cary, NC
Rodger RS (1975) The number of non-zero, post hoc contrasts from ANOVA and error rate I. Br J Math Stat Psychol 28:71–78
Schreiber SS, Hearse DJ, Oratz M, Rothschild MA (1977) Protein synthesis in prolonged cardiac arrest. J Mol Cell Cardiol 9:87–100
Vandenburgh HV, Kaufman S (1981) Stretch-induced growth of skeletal myotubes correlates with activation of the sodium pump. J Cell Physiol 109:205–214
Whalen WJ (1960) Some factors influencing O2 consumption of isolated cardiac muscle. Am J Physiol 198:1153–1156
Whalen WJ, Fangman J (1963) Respiration of heart muscle as affected by oxygen tension. Science 141:274–275
Wildenthal K, Griffin EE, Ingwall JS (1976) Hormonal control of cardiac protein and amino acid balance. Circ Res suppl 1, 38:I138–I142
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Loiselle, D.S. The rate of resting heat production of rat papillary muscle. Pflugers Arch. 405, 155–162 (1985). https://doi.org/10.1007/BF00584537
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00584537