Abstract
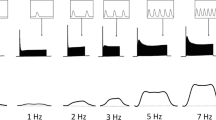
Peak force and membrane potential were recorded from papillary muscles and trabeculae excised from the ventricles of adult rat hearts. Experiments were performed at 2.5 mM Ca2+ and 26°C. In thick preparations (diameter 0.2–1.2 mm) an increase of stimulation frequency caused a reduction of peak force and action potential duration as has been found in many studies previously. In thin preparations (diameter <0.2 mm) both peak force and action potential duration were almost independent of stimulation frequency. When the flow of Tyrode solution through the muscle bath was reduced an increase of stimulation frequency caused a reduction of peak force and action potential duration in thin preparations. We conclude that the reduced peak force and action potential duration in papillary muscles at high stimulation frequencies is due to insufficient exchange of metabolites and oxygen between the medium and the core of the muscle. The results indicate that the critical diameter for the preparations is about 0.2 mm.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allen DG (1977) On the relationship between action potential duration and tension in cat papillary muscle. Cardiovasc Res 11:210–218
Allen DG, Kurihara S (1980) Calcium transients in mammalian ventricular muscle. Eur Heart J 1 (Suppl A):5–15
Alvarez J, Dorticos F, Morlans J (1981) Changes in electrical and mechanical activities of rabbit papillary muscle during hypoxic perfusion. J Physiol (Paris) 77:807–812
Boyett MR (1978) An analysis of the effect of the rate of stimulation and adrenaline on the duration of the cardiac action potential. Pflügers Arch 377:155–166
Boyett MR, Jewell BR (1978) A study of the factors responsible for rate-dependent shortening of the action potential in mammalian ventricular muscle. J Physiol (Lond) 285:359–380
Braveny P, Sumbera J (1970) Electromechanical correlations in the mammalian heart muscle. Pflügers Arch 319:36–48
Brutsaert DL (1967) Frequency potentiation and paired stimulation potentiation of cat papillary muscles in calcium and strontium containing media. Arch Int Physiol Biochim, 75:229–244
Edman KAP, Johannsson M (1976) The contractile state of rabbit papillary muscle in relation to stimulation frequency. J Physiol (Lond) 254:565–581
Forester GV, Mainwood GW (1974) Interval dependent inotropic effects in the rat myocardium and the effect of calcium. Pflügers Arch 352:189–196
Hoffmann BF, Kelly JJ (1959) Effects of rate and rhythm on contraction of rat papillary muscle. Am J Physiol 197:1199–1204
Jacobus WE, Pores IH, Lucas SK, Weisfeldt ML, Flaherty JT (1982) Intracellular acidosis and contractility in the normal and ischemic heart as examined by31PNMR. J Mol Cell Cardiol 14 (Suppl 3):13–20
Keung ECH, Aronson RS (1981) Non-uniform electrophysiological properties and electrotonic interaction in hypertrophied rat myocardium. Circ Res 49:150–158
Kurachi Y (1982) The effects of intracellular protons on the electrical activity of single ventricular cells. Pflügers Arch 394:264–270
Marshall RC, Nash WW, Shine KI, Phelps ME, Ricchiuti N (1981) Glucose metabolism during ischemia due to excessive oxygen demand or altered coronary flow in the isolated arterially perfused rabbit septum. Circ Res 49:640–648
Maylie JG (1982) Excitation-contraction coupling in neonatal and adult myocardium of cat. Am J Physiol 242:H 834-H 843
McCans JL, Lindenmayer GE, Munson RG, Evans RW, Schwartz A (1974) A dissociation of positive staircase (Bowditch) from ouabain-induced positive inotropism. Use of Verapamil. Circ Res 35:439–447
McDonald TF, Hunter EG, Macleod DP (1971) Adenosinetriphosphate partition in cardiac muscle with respect to transmembrane electrical activity. Pflügers Arch 322:95–108
Paradise NF, Schmitter JL, Surmitis JM (1981) Criteria for adequate oxygenation of isometric kitten papillary muscle. Am J Physiol 241:H 348-H 353
Rumberger E, Reichel H (1972) The force-frequency relationship: a comparative study between warm- and cold-blooded animals. Pflügers Arch 332:206–217
Schulze JJ (1981) Observations on the staircase phenomenon in guinea pig atrium. Pflügers Arch 391:9–16
Senges J, Brachmann DP, Rizos I, Kübler W (1981) Effect of glycolytic inhibitors on the sinoatrial node, atrium and atrioventricular node in the rabbit heart. J Mol Cell Cardiol 13:811–821
Siegl KS, McNeill JH (1982) Positive inotropic responses in cardiac muscles: influence of stimulation frequency and species. Can J Physiol Pharmacol 60:33–40
Sleator W, Furcgott RF, Gubareff T de, Krespi V (1964) Action potentials of guinea pig atria under conditions which alter contraction. Am J Physiol 206:270–282
Vleugels A, Carmeliet E, Bosteels S, Zaman M (1976) Differential effects of hypoxia with age on the chick embryonic heart. Changes in membrane potential intracellular K and Na, K efflux and glycogen. Pflügers Arch 365:159–166
Weiss J, Shine KI (1982) [K+]o accumulation and electrophysiological alterations during early myocardial ischemia. Am J Physiol 243:H 318-H 327
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schouten, V.J.A., ter Keurs, H.E.D.J. The force-frequency relationship in rat myocardium. Pflugers Arch. 407, 14–17 (1986). https://doi.org/10.1007/BF00580714
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00580714