Abstract
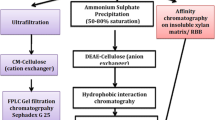
An extracellular xylanase enzyme fraction A from a mesophilicClostridium strain SAIV was purified by ammonium sulfate precipitation, Sephadex G-50 gel filtration and DEAE-Sephadex A-50 ion exchange. The xylanase exhibited a molecular weight of ≁30,000 and it was stable upto 55° C with an optimum temperature of 50° C. It was most stable between pH 5–7, with an optimum pH of around 6. The Km value was 7.0 mg·xylan ml-1 and Vmax was 36 μmol·xylose liberated mg-1 min-1. Carboxymethyl cellulose, filter paper cellulose and 4-p-nitrophenyl β-D-xylopyranoside were not hydrolysed. The specific activity of xylanase fraction A (9.8 U mg-1) is 2–10 fold higher than the specific activity of xylanase in other mesophilic, xylanolytic, obligate anaerobic bacteria. A minor fraction of xylanase activity designated as xylanase B was also obtained supporting the view that the multiplicity of xylanases is common in microorganisms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berenger JF, Frixon C, Bigliardi J & Creuzet N (1985) Production, purification, and properties of thermostable xylanase fromClostridium stercorarium. Can. J. Microbiol. 31: 635–643
Biely P (1985) Microbial xylanolytic systems. Trends Biotechnol. 3: 286–290
Debeire P, Priem B, Strecker G & Vignon M (1990) Purification and properties of an endo-1,4-xylanase excreted by a hydrolytic thermophilic anaerobe,Clostridium thermolacticum. Eur. J. Biochem. 187: 573–580
Dekker RFH & Richards GN (1976) Hemicellulases: their occurrence, purification, properties, and mode of action. Adv. Carbohydr. Chem. Biochem. 32: 277–352
Hespell RB, Wolf R & Bothast RJ (1987) Fermentation of xylans byButyrivibrio fibrisolvens and other ruminal bacteria. Appl. Environ. Microbiol. 53: 2849–2853
Laemmli UK (1970) Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature (London). 227: 680–685
Lee SF, Forsberg CW & Rattray JB (1987) Purification and characterization of two endoxylanases fromClostridium acetobutylicum ATCC 824. Appl. Environ. Microbiol. 53: 644–650
Lemmel SA, Datta R & Frankiewiez JR (1986) Fermentation of xylan byClostridium acetobutylicum. Enzyme Microb. Technol. 8: 217–221
Lowry OH, Rosebrough NJ, Farr AL & Randall RJ (1951) Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275
Miller GL (1959) Use of dinitrosalicylic acid agent for determination of reducing sugar. Anal. Chem. 31: 426–428
Murty MVS (1989) Studies on fermentation of major hemicellulosic components, Xylan and xylose by anaerobic bacteria. Ph.D. thesis, IIT Madras, India
Murty MVS & Chandra TS (1989) Isolation and characterization of xylose- and xylan-utilizing anaerobic bacteria. Antonie van Leeuwenhoek. 55: 153–163
— (1991) Expression of xylanase and cellulase enzymes in a newly isolatedClostridium sp. SAIV. Enzyme Microb. Technol. 13: 430–435
Pettipher GL & Latham MJ (1979) Production of enzymes degrading plant cell walls and fermentation of cellobiose byRuminococcus flavefaciens in batch and continuous culture. J. Gen. Microbiol. 110: 29–38
Reddy NR, Palmer JK, Pierson MD & Bothast RJ (1983) Wheat straw hemicelluloses; Composition and fermentation by human colonBacteroides, J. Agric. Food Chem. 31: 1308–1313
Reilly PJ (1981) Xylanases: Structure and function. Basic Life Sci. 18: 111–129
Saddler JN & Chan MKHE (1982) Optimisation ofClostridium thermocellum growth on cellulose and pretreated wood substrates. Appl. Microbiol. Biotechnol. 16: 99–104
Tanaka T, Shimomura Y, Himejima M, Taniguchi M & Oi S (1983) Characterization of xylan-utilizing anaerobes from mesophilic and thermophilic methane sludge and their sylan degrading enzymes. Agric. Biol. Chem. 50: 2185–2192
Uchino F & Nakane T (1981) A thermostable xylanse from a thermophilic acidophilicBacillus sp. Agric. Biol. Chem. 45: 1121–1127
Weber K & Osborn M (1969) Proteins and sodium dodecyl sulfate molecular weight determinations on polyacrylamide gels and related procedures. In: The Proteins. Vol. 1. Edited by H. Neurath, R.L. Hill & C. Boeder. Academic Press, Inc., N.Y. pp 179–233
Wong KKY, Tan LUL & Saddler JN (1988) Multiplicity of β-1,4-xylanase in microorganisms functions and applications. Microbiol. Rev. 52: 305–317
Woodward J (1984) Xylanases: Functions, properties and applications. Top. Enzyme Ferment. Biotechnol. 8: 9–30
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Murty, M.V.S., Chandra, T.S. Purification and properties of an extra cellular xylanase enzyme ofClostridium strain SAIV. Antonie van Leeuwenhoek 61, 35–41 (1992). https://doi.org/10.1007/BF00572120
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00572120