Summary
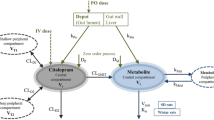
The time courses of plasma metoclopramide concentrations were followed in six subjects after oral and intravenous single dose administration. Plasma concentration-time data following i.v. administration in each subject were found to fit a two compartment model with a mean terminal half-life of 4.55 h±0.80 h and a mean distribution half-time of 0.35 h±0.09 h. Volumes of distribution were high (3.43±1.181 · kg−1), and clearances (0.53±0.191 · kg−1h−1) approached liver plasma flow. This suggests that metoclopramide occurs at higher concentrations in tissues than in plasma, and that its clearance is probably limited by liver blood flow rather than liver metabolic capacity. The post-absorption decline in metoclopramide plasma levels after oral administration was also biexponential in each subject. The terminal half-life was 5.17 h±0.98 h. Mean volume of distribution and mean clearance were similar to intravenous values (after adjustment for bioavailability). Oral absorption was rapid with peak plasma concentrations being reached at a mean time of 0.93 h. A mean bioavailability of 0.77 was calculated for the six subjects, and it was postulated that this incomplete availability is due to a first-pass effect. The inter-individual variation in the degree of ‘first-pass’ was considerable (0.47–1.14).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arita T, Hori R, Ito K, Ichikawa K, Uesugi T (1970) Transformation and excretion of drugs in biological systems. II. Transformation of metoclopramide in rabbits. Chem Pharm Bull 18: 1663–1669
Bakke OM, Segura J (1976) The absorption and elimination of metoclopramide in three animal species. J Pharm Pharmacol 28: 32–39
Bateman DN, Kahn C, Mashiter K, Davies DS (1978) Pharmacokinetics and concentration effect studies with intravenous metoclopramide. Br J Clin Pharmacol 6: 401–407
Bateman DN, Kahn C, Davies DS (1979) Concentration effect studies with oral metoclopramide. Br J Clin Pharmacol 8: 179–182
Beckett AH, Huizing G (1975) In vitro metabolism of metoclopramide. J Pharm Pharmacol 27: (Suppl) 42P
Chiou WL (1975) Estimation of hepatic first-pass effect of acetaminophen in humans after oral administration. J Pharm Sci 64: 1734–1735
Gibaldi M, Perrier D (1975) Pharmacokinetics. Marcel Dekker, New York
Graffner C, Lagerstrom P, Lundborg P (1979) Pharmacokinetics of metoclopramide intravenously and orally determined by liquid chromatography. Br J Clin Pharmacol 8: 469–474
Knott GD (1979) MLAB - a mathematical modelling tool. Comput Programs Biomed, 10: 271–280
Matts SG (1974) Metoclopramide in the treatment of migraine. Practitioner 212: 887–890
McCallum RW, Ippoliti AF, Cooney C, Sturdevant RAL (1977) A double-blind trial of metoclopramide in symptomatic gastrooesophageal reflux. Gastroenterology 70: 354–357
Ross-Lee LM, Eadie MJ, Bochner F, Hooper WD, Tyrer JH (1980) Electron capture gas chromatographic assay for metoclopramide in plasma. J Chromatogr 183: 175–184
Sedman AJ, Wagner JG (1976) C-STRIP, a Fortran IV computer program for obtaining initial polyexponential parameter estimates. J Pharm Sci 65: 1006–1010
Tam YK, Axelson R (1978) Sensitive electron-capture GLC determination of metoclopramide in biological fluids. J Pharm Sci 67: 1073–1077
Teng L, Bruce R, Dunning L (1977) Metoclopramide metabolism, and determination by high pressure liquid chromatography. J Pharm Sci 66: 1615–1618
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ross-Lee, L.M., Eadie, M.J., Hooper, W.D. et al. Single-dose pharmacokinetics of metoclopramide. Eur J Clin Pharmacol 20, 465–471 (1981). https://doi.org/10.1007/BF00542101
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00542101