Summary
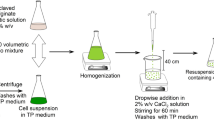
Plant cells, entrapped in agarose or alginate, were permeabilized in a new procedure that retain cell viability, permitting the possibility of reusing biomass after release of intracellular products. Dimethylsulfoxide (DMSO) was used to make the cells permeable. The activity of isocitrate dehydrogenase, an intracellular enzyme, was used as an indicator of plasma membrane permeability. Cells from three plant species require different concentrations of DMSO for complete permeabilization (i.e., for maximal isocitrate dehydrogenase activity). Cells of Catharanthus roseus permeabilized with up to 5% DMSO remained viable, and released 85–90% of the intracellularly stored products (ajmalicine isomers). In model production systems, C. roseus cells entrapped in agarose or alginate beads were intermittently permeabilized for release of products in a cyclic process. An increase in product yield was observed for each cycle because of increase in biomass (cell growth) within the beads.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bajaj YPS (1976) Regeneration of plants from cell suspensions frozen at −20, −70, and −196° C. Physiol Plant 37:263–268
Barz W, Reinhard E, Zenk MH (eds). (1977) Plant tissue culture and its biotechnological application. Springer, Berlin Heidelberg New York
Brodelius P, Deus B, Mosbach K, Zenk MH (1979) Immobilized plant cells for the production and transformation of natural products. FEBS Lett 103:93–97
Brodelius P, Deus B, Mosbach K, Zenk MH (1980) The potential use of immobilized plant cells for the production and transformation of natural products. In: Weetall HH, Royer GP (eds) Enzyme engineering, vol 5. Plenum Press, New York, pp 373–381
Brodelius P, Nilsson K (1980) Entrapment of plant cells in different matrices. A comparative study. FEBS Lett 122:312–316
Brodelius P, Mosbach K (1982) Immobilized plant cells. In: Laskin AI (ed) Advances in applied microbiology, vol 28. Academic Press, New York, pp 1–26
Brodelius P (1983) Immobilized plant cells. In: Mattiasson B (ed) Immobilized cells and organelles. CRC Press, Boca Raton (in press)
Deus B, Zenk MH (1982) Exploitation of plant cells for the production of natural compounds. Biotechnol Bioeng 24:1965–1974
Dougall DK, Wetherell DF (1974) Storage of wild carrot cultures in the frozen state. Cryobiology 11:410–415
Felix H, Brodelius P, Mosbach K (1981) Enzyme activities of the primary and secondary metabolism of simultaneously permeabilized and immobilized plant cells. Anal Biochem 116:462–470
Felix H, Mosbach K (1982) Enhanced stability of enzymes in permeabilized and immobilized cells. Biotechnol Lett 4:181–186
Kurz WGW, Constabel F (1979) Plant cell cultures a potential source of pharmaceuticals. In: Perlmann D (ed) Advances in applied microbiology, vol 25. Academic Press, New York, pp 209–240
Linsmaier EM, Skoog F (1965) Organic growth factor requirements of tobacco tissue cultures. Physiol Plant 18:100–127
Nilsson K, Birnbaum S, Flygare S, Linse L, Schroder U, Jeppsson U, Larsson P-O, Mosbach K, Brodelius P (1983) A general method for the immobilization of cells with preserved viability. Eur J Appl Microbiol Biotechnol 17:319–326
Ogino T, Hiraoka N, Tabata M (1978) Selection of high nicotine-producing cell lines of tobacco callus by single-cell cloning. Phytochemistry 17:1907–1910
Schenk RU, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50:199–204
Staba EJ (ed) (1980) Plant tissue culture as a source of biochemicals. CRC Press, Boca Raton
Veliky IA, Rose D (1973) Nitrate and ammonium nitrogen nutrients for plant cell cultures. Can J Bot 51:1837–1844
Widholm JM (1972) The use of fluorecein diacetate and phenosafranine for determining viability of cultured plant cells. Stain Technol 47:189–194
Yamamoto Y, Mizuguchi R, Yamada Y (1982) Selection of a high and stable pigment-producing strain in cultured Euphorbia millii cells. Theor Appl Genet 61:113–116
Zenk MH, El-shagi H, Arens H, Stockigt J, Weiler EW, Deus B (1977) Formation of the indole alkaloids serpentine and ajmalicine in cell suspension cultures of Catharanthus roseus. In: Barz W, Reinhard E, Zenk MH (eds) Plant tissue cultures and its bio-technological application. Springer, Berlin Heidelberg New York, pp 27–43
Zenk MH (1978) The impact of plant tissue culture on industry. In: Thorpe TA (ed) Frontiers of plant tissue culture. University of Calgary Press, Calgary, pp 1–17
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brodelius, P., Nilsson, K. Permeabilization of immobilized plant cells, resulting in release of intracellularly stored products with preserved cell viability. European J. Appl. Microbiol. Biotechnol. 17, 275–280 (1983). https://doi.org/10.1007/BF00508020
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00508020