Summary
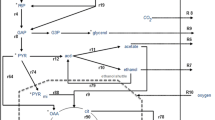
The kinetics of glucose and xylose utilization by batch cultures of Candida utilis were studied under aerobic and anaerobic conditions during growth in complex media. Rapid ethanol formation occurred during growth on glucose when aerobic cultures were shifted to anaerobic conditions. However, with xylose as a substrate, transfer to anaerobiosis resulted in an immediate cessation of metabolic activity, as evidenced by the absence of both ethanol formation and xylose utilization. The inability of the yeast to ferment xylose anaerobically was not due to the absence of key enzymes of the fermentation pathway, since the addition of glucose to such cultures resulted in the immediate conversion of glucose to ethanol. Furthermore, when the enzyme xylose isomerase was added to an anaerobic xylose culture, immediate conversion of xylose to ethanol was observed. This indicates that the inability of the yeast to form ethanol from xylose under anaerobic conditions is caused by metabolic events associated with the conversion of xylose to xylulose. A hypothesis is put forward which explains that ethanol production from xylose by yeast under anaerobic conditions is negligible. It is suggested that the failure to ferment xylose anaerobically is due to a discrepancy between production and consumption of NADH in the overall conversion of xylose to ethanol. When a hydrogen acceptor (i.e. acetoin) was added to anaerobic cultures of C. utilis, xylose utilization resumed, and ethanol and acetate were produced with the concomitant stoicheiometric reduction of acetoin to 2,3-butanediol.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barnett JA, Payne RW, Yarrow D (1979) A guide to identifying and classifying yeasts. Cambridge University Press, London
Bruinenberg PM, van Dijken JP, Scheffers WA (1983a) A theoretical analysis of NADPH production and consumption in yeasts. J Gen Microbiol 129:953–964
Bruinenberg PM, van Dijken JP, Scheffers WA (1983b) An enzymic analysis of NADPH production and consumption in Candida utilis. J Gen Microbiol 129:965–971.
Chakravorty M, Veiga LA, Bacila M, Horecker BL (1962) Pentose metabolism in Candida II. The diphosphopyridine nucleotide-specific polyol dehydrogenase of Candida utilis. J Biol Chem 237:1014–1020
Dekker RFH (1982) Ethanol production from D-xylose and other sugars by the yeast Pachysolen tannophilus. Biotechnol Lett 4:411–416
Dellweg H, Debus D, Methner H, Schulze D, Saschewag I (1982) Xylose-Vergärung mit Pachysolen tannophilus. In: Dellweg H (Hrsg) 5. Symposium Technische Mikrobiologic, Berlin. Inst Gärungsgewerbe u Biotechnol, Berlin, S 200–207
Fiechter A, Fuhrmann GF, Käppeli O (1981) Regulation of glucose metabolism in growing yeast cells. Adv Microb Physiol 22: 123–183
Gong CS, Chen LF, Flickinger MC, Chiang LC, Tsao GT (1981) Production of ethanol from D-xylose by using D-xylose isomerase and yeasts. Appl Environ Microbiol 41:430–436
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. In: Norris JR, Ribbons DW (eds) Meth microbiol 5B. Academic Press, London New York, pp 209–344
Horecker BL (1962) Pentose metabolism in bacteria. John Wiley and Sons Inc, New York London
Jeffries TW (1981) Conversion of xylose to ethanol under aerobic conditions by Candida tropicalis. Biotechnol Lett 3:213–218
Kluyver AJ (1914) Biochemische suikerbepalingen. PhD Thesis, Delft University of Technology, Delft, The Netherlands, p 17
Margaritis A, Bajpai P (1982) Direct fermentation of D-xylose to ethanol by Kluyveromyces marxianus strains. Appl Environ Microbiol 44:1039–1041
Oura E (1977) Reaction products of yeast fermentation. Proc Biochem 12 (3): 19–21, 35
Rosenberg SL (1980) Fermentation of pentose sugars to ethanol and other neutral products by microorganisms. Enzyme Microb Technol 2:185–193
Scheffers WA (1966) Stimulation of fermentation in yeasts by acetoin and oxygen. Nature 210:533–534
Scheffers WA, Nanninga GL (1977) The anaerobic inhibition of fermentation (Custers effect) in Brettanomyces. EUCHEM conference on metabolic reactions in the yeast cell in anaerobic and aerobic conditions. Helsinki, pp 53–55
Scheffers WA, Wikén TO (1969) The Custers effect (negative Pasteur effect) as a diagnostic criterion for the genus Brettanomyces. Ant v Leeuwenhoek 35 Suppl yeast symp: A31–A32
Scheffers WA, van Dijken JP, Kaytan G, Kloosterman M, Wijsman MR, van Kleeff BHA (1982) Effect of oxygen on growth, alcohol, acetic acid, and glycerol production by the yeasts Brettanomyces intermedius and Zygosaccharomyces bailii. In: Dellweg H (Hrsg) 5. Symp Technische Mikrobiologie. Inst Gärungsgewerbe Biotechnol, Berlin, S 213–220
Schneider H, Wang PY, Chan YK, Maleszka R (1981) Conversion of D-xylose into ethanol by the yeast Pachysolen tannophilus. Biotechnol Lett 3:89–92
Sims AP, Barnett JA (1978) The requirement of oxygen for the utilization of maltose, cellobiose and D-galactose by certain anaerobically fermenting yeasts (Kluyver effect). J Gen Microbiol 106:277–288
Slininger PL, Bothast RJ, van Cauwenberge JE, Kurtzman CP (1982) Conversion of D-xylose to ethanol by the yeast Pachysolen tannophilus. Biotechnol Bioeng 24:371–384
Smiley KL, Bolen PL (1982) Demonstration of D-xylose reductase and D-xylitol dehydrogenase in Pachysolen tannophilus. Biotechnol Lett 4:607–610
Suihko ML, Dražić M (1983) Pentose fermentation by yeasts. Biotechnol Lett 5: 107–112
Tomoyeda M, Horitsu H (1964) Pentose metabolism by Candida utilis. Part I. Xylose isomerase. Agr Biol Chem 28:139–143
Van Tilburg R (1983) Engineering aspects of biocatalysts in industrial starch conversion technology. PhD Thesis, Delft University of Technology, Delft, The Netherlands
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bruinenberg, P.M., de Bot, P.H.M., van Dijken, J.P. et al. The role of redox balances in the anaerobic fermentation of xylose by yeasts. European J. Appl. Microbiol. Biotechnol. 18, 287–292 (1983). https://doi.org/10.1007/BF00500493
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00500493